These special features and the roundtable meeting were sponsored and moderated by Takeda.
Authors are Paid Consultants of Takeda.
All trademarks are the property of their respective owners. ©2020 Millennium Pharmaceuticals, Inc.,
a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
CPS1435
How are Blood Cancers Different from Other Cancers?
History of CML and its Treatment with TKIs
Living with CML: The “New Normal”
Resistant CML – “Like Being Diagnosed All Over Again”
PCR Testing as a CML Monitoring Tool
Over the past 20 years, advances in the treatment of chronic myeloid leukemia (CML) have made this type of cancer highly treatable, such that most patients can live long and productive lives. Today, people diagnosed with CML who are treated with targeted medications have similar life expectancies as people without CML.1 This big change in outcomes for people with CML can be largely attributed to the discovery of targeted therapies known as tyrosine kinase inhibitors (TKIs). These agents are pills that are taken once or twice a day. TKIs, which first became available in the United States in 2001,2 have revolutionized the treatment of CML.3
However, not all CML is the same. Although most people with CML have excellent outcomes, up to 1 in 4 patients will become resistant to their first drug treatment and up to 1 in 5 will have side effects that prevent them from continuing on initial therapy (Figure 1).4-8 When CML progresses after treatment or does not respond to treatment, it is called resistant CML.9
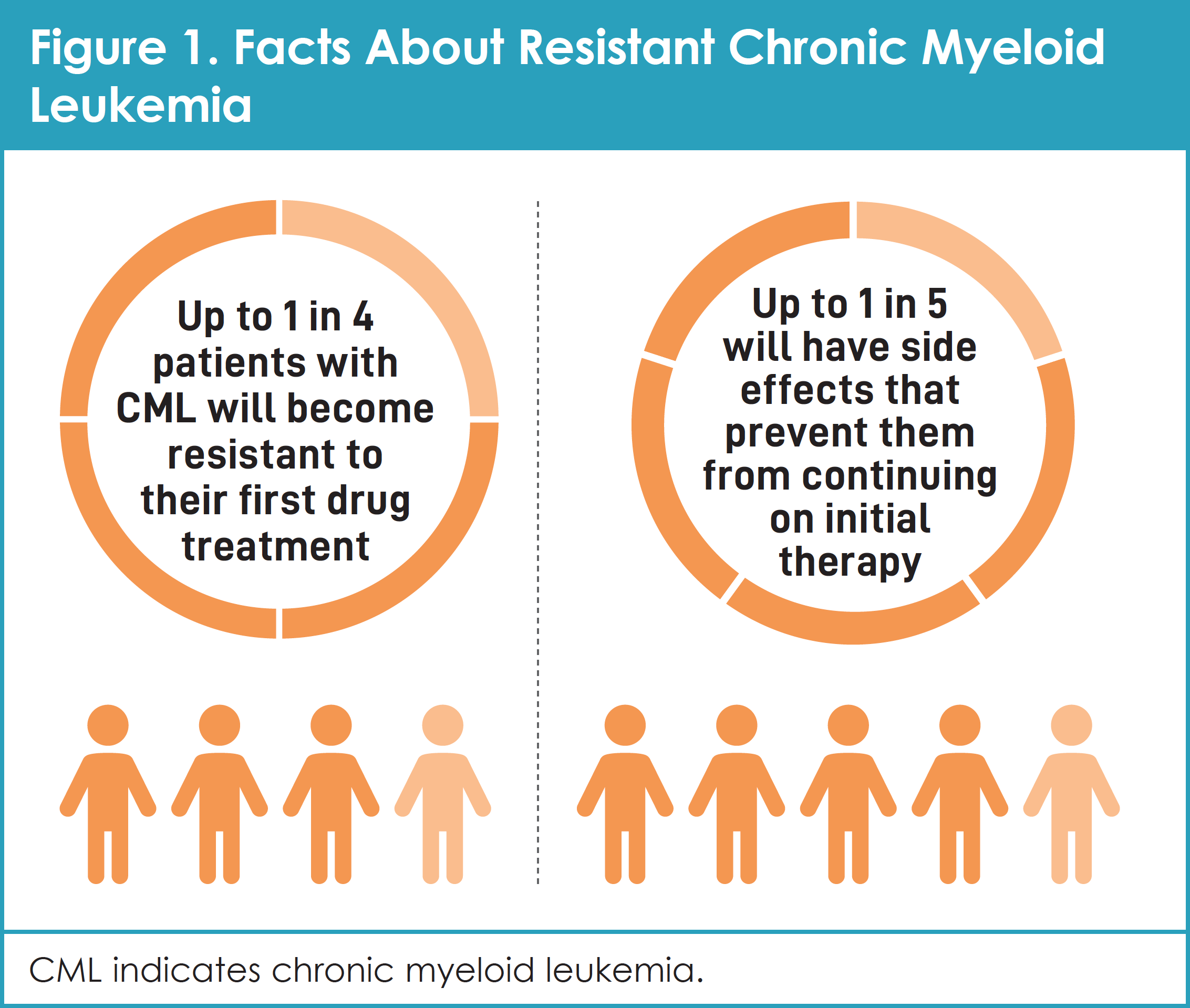
This article reviews key developments in the treatment of patients with CML, including resistant CML. It provides patients and their caregivers with knowledge that can help them partner effectively with their medical team. The ideas and recommendations come from a group of people living with the disease. In June 2019, 6 patients with CML and 1 caregiver met in Cambridge, Massachusetts, to take part in the CML Roundtable, which was sponsored by Takeda Oncology (Sidebar). Roundtable participants shared their own experiences with initial treatment of CML and, in some cases, their experiences with resistant CML, to illustrate the unique needs of patients who live with this disease.
Roundtable Participants
Dean (New Orleans, LA). Dean is married with 2 children. He was diagnosed with CML in December 2003 and has been enrolled in multiple clinical trials of TKIs. Dean is part of the Leukemia & Lymphoma Society advocacy leadership committee and is active in Facebook support groups for patients with CML.
Michele (Boise, ID). Michele is married with 3 children. She was diagnosed with CML in February 2011 and has received multiple treatments for CML. A journalist, Michele has developed her own community of patients with CML through her blog and Facebook group.
Hans (Woodinville, WA). Hans is married with 4 children. He was diagnosed with CML in November 2008 and has undergone multiple treatments for the disease. Hans is active in the CML community, participating in interviews, videos, support groups, his blog, and a Facebook group.
Justin (Grand Prairie, TX). Justin is married and owns a digital marketing company. He was diagnosed with CML in January 2006 at 24 years of age and has received several treatments. Justin is an active member of the CML community, leading support groups and participating in fundraising events and patient panels.
Karen (Eden Prairie, MN). Karen, a breast cancer survivor, was diagnosed with CML in 2005 and has received several treatments and participated in clinical trials. She successfully advocated for drug access with the Food and Drug Administration when one of the available TKI treatments was temporarily withdrawn from the market.
Joannie (Costa Mesa, CA). Joannie is married with 3 children, 5 grandchildren, and 5 great-grandchildren. She was diagnosed with CML in 2001. Her condition remains stable on the same drug that she first received 18 years ago. She is patient-centric, and the founder, leader, and organizer of a long-standing CML support group. Joannie is also a patient advocate for drug accessibility and sustainable affordability, as well as a fundraiser for efforts to find cures for blood cancers. Joannie is an officer on the Board of Trustees of the Leukemia & Lymphoma Society.
Jerry (Costa Mesa, CA). Jerry is married to Joannie. Together, they provide CML-specific patient outreach in the United States through the CML Buster Foundation and CML Busters Family Support Group. Jerry and his wife also support patients with CML internationally through their partnership with the CML Advocates Network. Jerry has concentrated his outreach efforts on the support of caregivers (family, friends, and medical professionals) to the CML community since 2003 and has presented internationally on the subject. He also works to raise awareness and funds for CML research, innovation, and treatment access.
Melvin (Jonesboro, GA). Melvin is married with 1 child. He was diagnosed with CML in 1995 and participated in a clinical trial of the first TKI. Since then, his condition remains stable on the same drug that he first received more than 20 years ago. Melvin is engaged in the CML community and works with the Leukemia & Lymphoma Society to inform and support other people with CML.
This publication discusses how CML is diagnosed, how it is different from other types of blood cancers, the important role of TKIs in the management of CML, and how doctors use special tests and monitoring strategies to choose the best course of treatment for their patients. This article also contains helpful insights and practical advice from individuals with CML who participated in the Roundtable meeting, as well as a list of valuable resources and services for patients and their caregivers.
What is CML?
CML is a type of cancer that starts from cells found in bone marrow.10 Healthy bone-marrow cells grow into different, more refined types of blood cells. In CML, these bone-marrow cells grow out of control.9
BCR-ABL, a genetic mutation, is found in almost all patients with CML. BCR-ABL is formed by the combination of 2 genes, known as BCR and ABL. It is sometimes called a fusion gene. BCR is normally on chromosome number 22, whereas ABL is normally on chromosome number 9. The BCR-ABL mutation happens when pieces of BCR and ABL genes break off and fuse together. The mutated chromosome 22 is called the Philadelphia chromosome after the city where researchers first discovered it.11
The BCR-ABL gene is not the type of mutation that is inherited from your parents. It is a “somatic” mutation, which means it happens later in life.
In CML, there is often a high number of white blood cells (WBCs), more specifically known as neutrophils and granulocytes. The body needs these cells to fight infections. Ordinarily, more WBCs are made during infections or times of stress, and then the number of WBCs returns to normal when the infection is gone. In CML, the abnormal protein called BCR-ABL acts like a switch that is stuck in the “on” position, so that too many WBCs grow and multiply. In CML, the body may also make too many blood platelets, which help the blood to clot. Finally, the body may also make too few red blood cells (RBCs), which carry oxygen.9 People who have too few RBCs, a condition also known as anemia, can feel tired, can be short of breath, and/or may look pale (Figure 2).
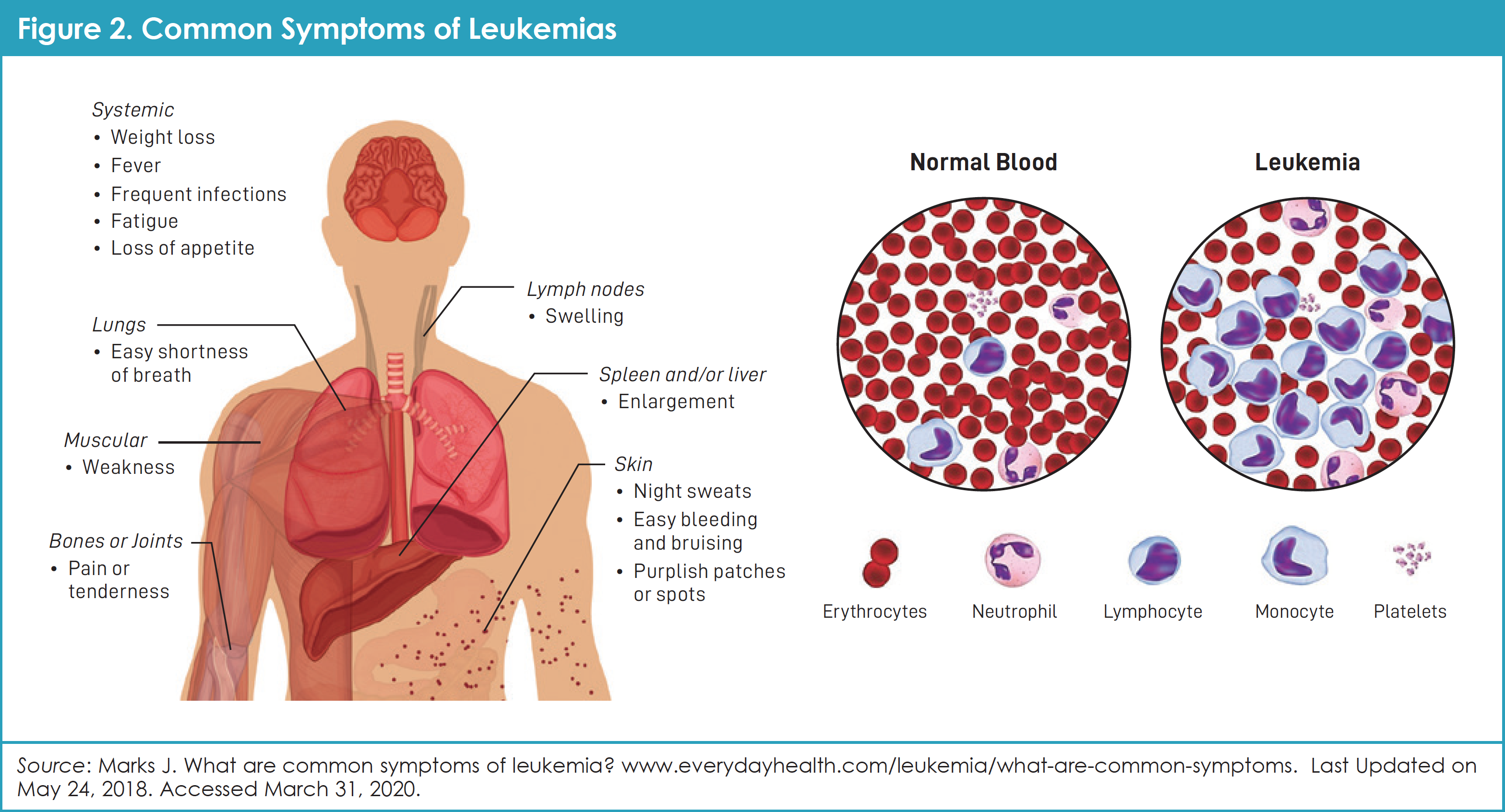
CML is divided into 3 different phases: chronic phase, accelerated phase, and blast phase.9
- Chronic-phase CML: In this early phase of CML, the blood and bone marrow contain less than 10% of immature WBCs or blast cells. This phase can last for several years with effective treatment. Without effective treatment, chronic-phase CML can progress to the accelerated or blast phase. Approximately 90% of people have chronic-phase CML when they are diagnosed. Some people with chronic-phase CML have symptoms when they are diagnosed, whereas others do not. Most symptoms go away with treatment.9
- Accelerated-phase CML: Although accelerated phase has no single definition, most patients in this phase of CML have 10% to 19% blasts (immature white blood cells) in both the blood and bone marrow, or more than 20% basophils (a special type of WBC) in the blood. CML cells sometimes have new cytogenetic changes in addition to the Philadelphia chromosome. This happens because of additional DNA damage and mutations.9
- Blast-phase CML or blast crisis: Patients in this phase have 20% or more blasts in the blood or bone marrow. It is difficult to control the number of WBCs; as a result, WBC counts can get very high. CML cells often have additional genetic changes, and blast cells can look like cells that are seen in patients with acute leukemia (acute myeloid leukemia for most patients, and acute lymphocytic leukemia for approximately 25% of patients in blast crisis). In this phase, patients often have fever, an enlarged spleen, and weight loss, and they feel poorly.9
How are Blood Cancers Different from Other Cancers?
Cancer is defined as abnormal growth of cells. Cancers can occur in an organ or body structure or in the blood or bone marrow. In an organ or body structure, cancer cells multiply to the point at which a mass of them is large enough to be noticed by the patient or doctor.
Cancers such as CML that do not grow into a mass or tumor are called “blood cancers” or “liquid tumors.” These include cancers of the blood and bone marrow, which are also known as lymphomas and leukemias. Blood cancers are often found by doctors after reviewing laboratory tests. More than 1 million cancer cells must be present before they can be detected in the blood.12
When an organ or body structure is affected by cancer, the tumor usually must reach a size of 1 centimeter or contain 1 million cells before it can be felt or seen. These cancers are called “solid tumors.” Examples of common solid tumors include cancers of the lung, breast, prostate, colon, and bladder.12
Figure 3 shows how CML is different from other blood cancers.13
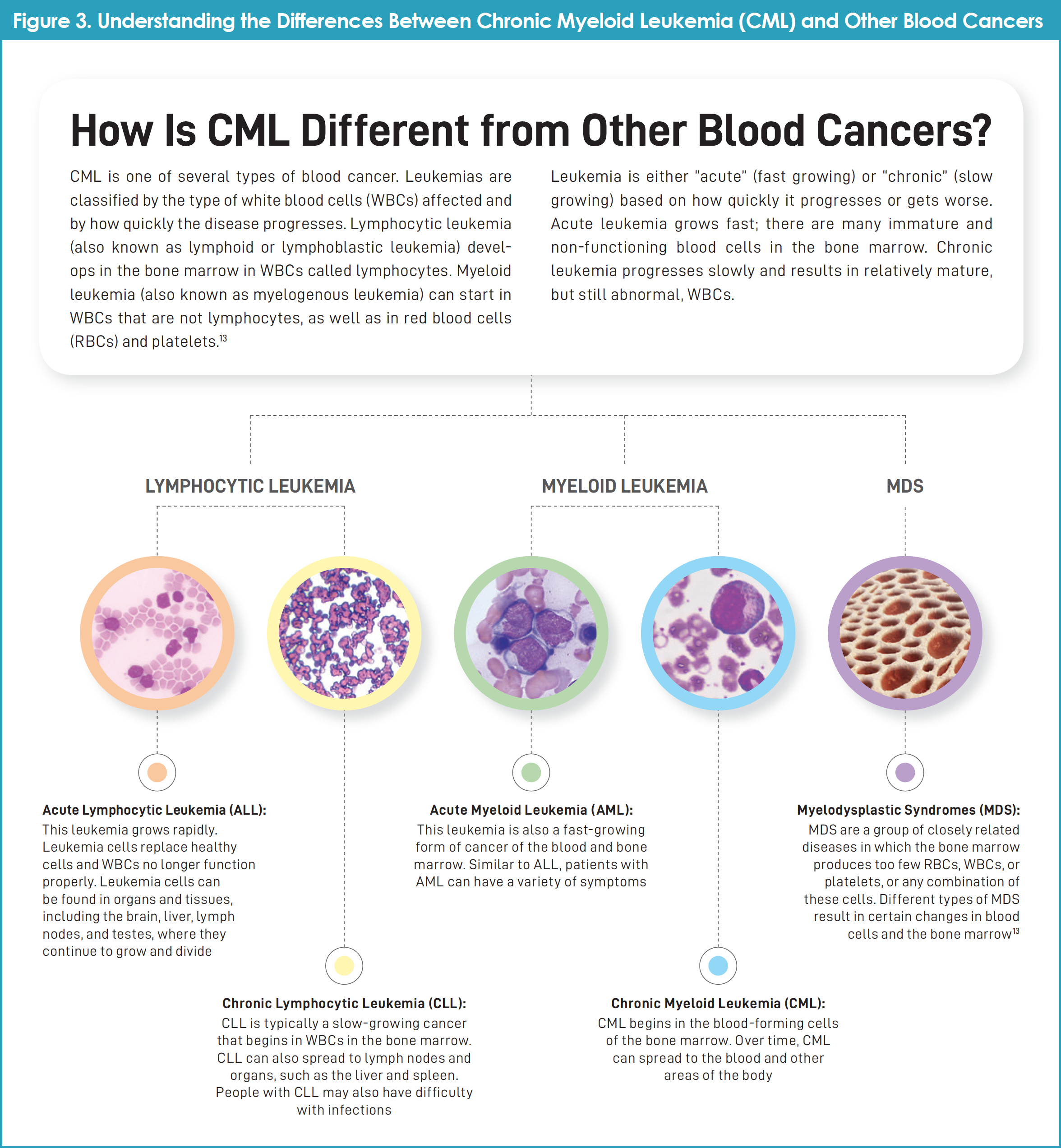
History of CML and its Treatment with TKIs
The treatment of CML has been revolutionized by the development of imatinib and other TKIs. Key events in the discovery of new treatments for CML are as follows14,15:
1960: After 2 Philadelphia-based doctors, Nowell and Hungerford, looked at genetic material from cancer cells of patients with CML, they noticed that 1 of these patients’ 46 chromosomes was abnormally short. The short chromosome was later named “the Philadelphia chromosome.” Researchers now know that 95% of patients with CML have the Philadelphia chromosome (also called Ph-positive or Ph+)
1970s: Researchers discovered that the Philadelphia chromosome forms when 2 chromosomes break and these pieces merge together into what is now known as a fusion gene called BCR-ABL
1990s: Druker and Lydon discovered that a drug called imatinib can block the activity of BCR-ABL
1998: Druker and colleagues tested imatinib in a clinical trial of patients with CML. The drug resulted in disappearance of the cancer for most of the patients whose CML was in the early or “chronic” phase
2003+: Follow-up data showed that almost all patients with CML who participated in clinical trials of new TKIs remained in remission with acceptable quality of life and generally manageable side effects14
Today: Data from clinical trials show that someone with CML who is in remission after 2 years of treatment with a TKI may have the same life expectancy as someone of the same age who does not have cancer.14
Imatinib and the other drugs that target BCR-ABL are called tyrosine kinase inhibitors (TKIs). TKIs are different from chemotherapy drugs, in part because these agents can be taken by mouth instead of being given by injection. Because TKIs can specifically target the BCR-ABL enzyme, they have different side effects compared with chemotherapy.
“People who are afraid of chemotherapy have to realize that today things are different. This isn’t your grandfather’s CML!” – Dean
Use of TKIs is now the standard treatment for CML, particularly for patients whose disease is in the chronic phase. TKIs can also help patients with more advanced CML, which is called accelerated- or blast-phase CML.16 In the United States there are a number of TKIs approved for the treatment of patients with CML.
For most people with CML, treatment must be taken each day until it no longer works or results in side effects that are unacceptable to the patient. For some people with CML who have long-lasting and deep responses to a TKI, it may be possible to stop taking the TKI or to lower the dose.16
Initial Diagnosis of CML
At the time of diagnosis, people with CML may or may not experience signs and symptoms. Sometimes, a patient’s symptoms are related to a medical condition other than CML. Recall that CML typically progresses slowly, meaning symptoms are often mild at first. These symptoms can get worse over time, but this will happen gradually.9
Examples of signs or symptoms that a patient with CML may feel include the following9:
- Fatigue
- Weakness, including shortness of breath while doing everyday activities
- Fever
- Excessive sweating, especially at night
- Weight loss
- Abdominal swelling or discomfort due to an enlarged spleen
- Feeling full even if the person has not eaten much
- Itching
- Bone pain
- Bruising
- Bleeding.
Doctors use many types of tests to diagnose leukemia, including CML. Tests also help doctors to learn more about the extent of the disease. Doctors can also perform tests to learn which treatments could work best.9
For most people with CML, the Philadelphia chromosome and the BCR-ABL fusion gene can be found through molecular testing. In addition to a bone-marrow biopsy, detection of the BCR-ABL fusion gene confirms the diagnosis of CML. Polymerase chain reaction (PCR) testing is also used to monitor how well treatment for CML is working. Specifically, PCR testing determines whether treatment with a TKI is effectively lowering the number of cells with the Philadelphia chromosome.9
The immediate goal of treatment for CML is to reduce the number of leukemia cells, which typically reduces symptoms of the disease (Figure 4: Early Treatment Response Milestones to TKI Therapy in Chronic Phase CML recommended by National Comprehensive Cancer Network [NCCN]). Longer-term goals are to decrease or eliminate cells with the Philadelphia chromosome. This slows down or prevents CML from moving to the blast phase.9,17
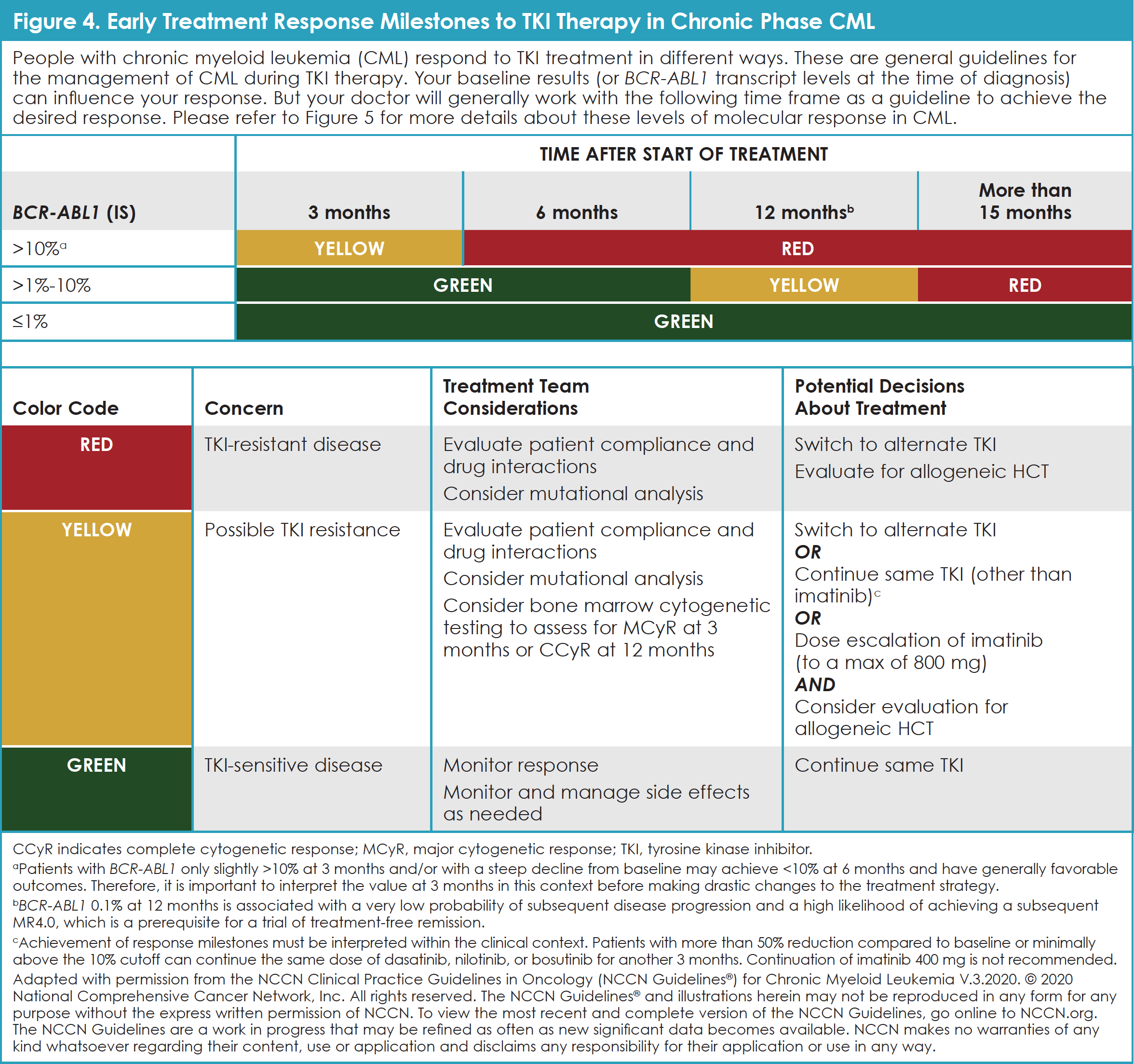
At the time of diagnosis, doctors plan treatment and predict “prognosis,” or the chance of recovery based on the phase of CML. General treatment options for people who are diagnosed with CML include TKIs; chemotherapy, such as hydroxyurea and omacetaxine mepesuccinate; and interferon. For patients with chronic-phase CML, TKIs are the standard first treatment. Bone-marrow transplant, also called stem-cell transplant, is often considered for select patients with chronic-phase CML whose disease no longer responds to TKI therapy.9
Although a detailed review of treatment options and their risks and benefits is beyond the scope of this article, readers can learn more about CML at Cancer.Net (www.cancer.net/cancer-types/leukemia-chronic-myeloid-cml), the Leukemia & Lymphoma Society (www.lls.org), and other patient-friendly websites that have been developed by trusted sources. A list of these resources is provided at the end of this article.
Patients and caregivers who participated in the Takeda CML Patient Roundtable emphasized that people with CML should understand all of the available treatment options for their disease, and actively participate in the decision-making process with their healthcare team.
Living with CML: The “New Normal”
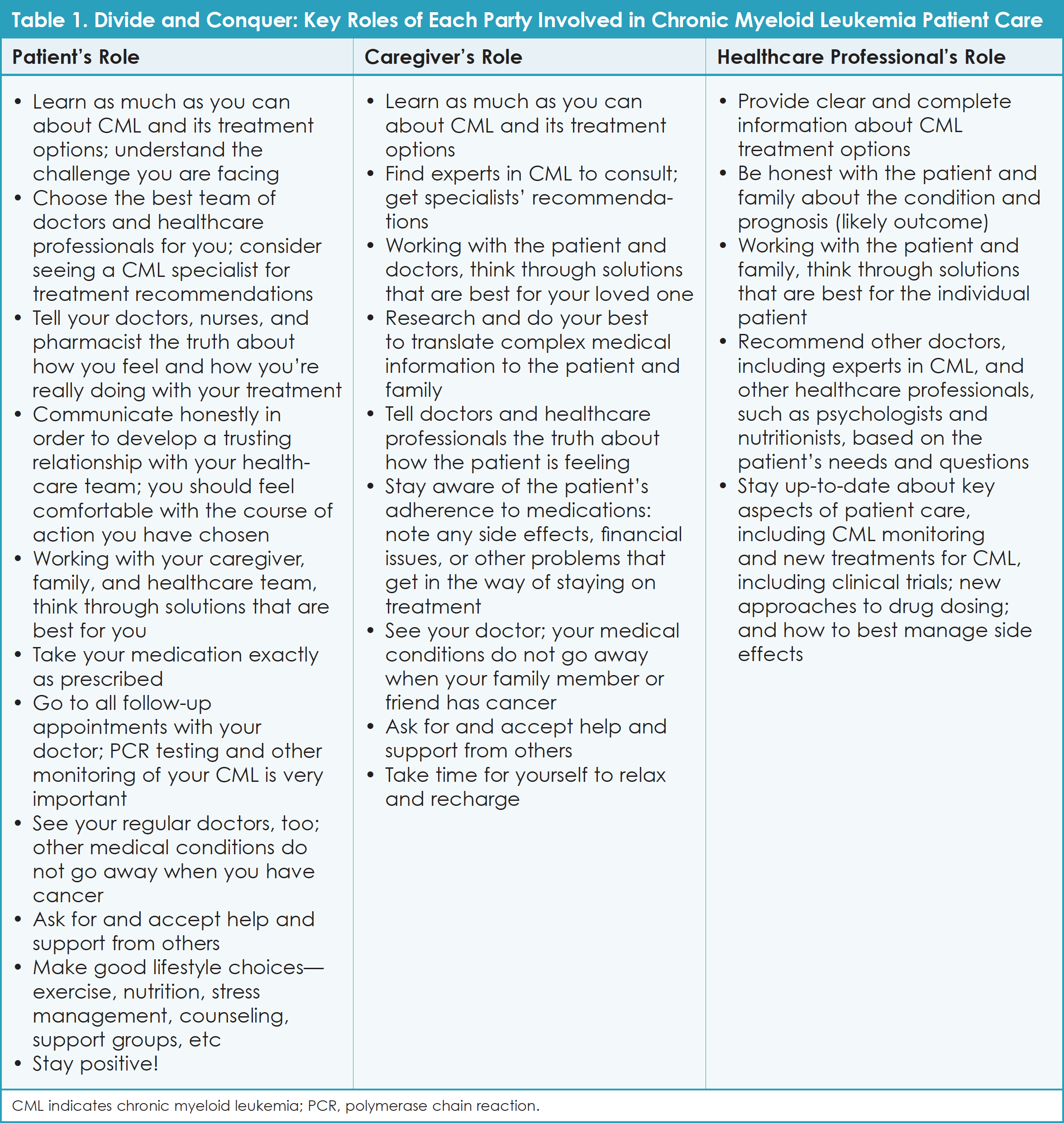
The patients and caregivers who participated in the Takeda CML Patient Roundtable discussion agreed that living with CML has its ups and downs, but that this cancer has become a part of their “new normal.” Table 1 describes key tasks that each party—patients, caregivers, and healthcare professionals—should take on after someone has been diagnosed with CML. These ideas are based on insights from Roundtable participants and from Cancer.Net.9
“We [as people with CML] don’t know what we don’t know. We think all oncologists know about all types of cancer, but they cannot be experts in everything. You see that knowledge gap only after seeing a CML specialist.” – Justin
“I researched every drug for CML. I doctor shopped. I went through 4 doctors before finding one that fit me. A lot of people with cancer go to 1 doctor, follow their instructions, and don’t question them. Advocate for yourself!” – Michele
“Patients need to be hopeful and remain hopeful. If they have confidence in their doctors, it helps.” – Jerry
“This is your life, your disease. You are allowed to ask questions. Don’t be afraid to push back or seek others’ advice.
Use your voice!” – Hans
Top Questions to Ask Your Doctor About Resistant CML
- What is the best treatment for me based on my test results, including genetic testing that can identify mutations associated with CML?
- What will this treatment do for me? Does it get rid of leukemia cells? Does it help me feel better? Will I be cured?
- How will this treatment affect my daily life? Will I be able to work, exercise, and do my usual activities? How can I prevent and/or manage side effects?
- What follow-up tests will I need? How often should I be tested? What results should I expect? Are there guidelines that I should become familiar with?
- What is the risk that my CML will worsen? Are there signs and symptoms I should watch for?
- What resources are available to help me learn more about resistant CML and find support services?
Please see Cancer.Net for a comprehensive list of questions to ask your doctor or other members of the healthcare team to help you better understand your CML diagnosis, treatment plan, and monitoring.9
Resistant CML – “Like Being Diagnosed All Over Again”
If CML has come back after treatment or does not respond to treatment, it is called resistant CML.9 Based on reviews of findings from clinical trials, up to 40% of patients with chronic-phase CML may become resistant to first-line imatinib.18
“I was just crushed when my CML relapsed. It was sneaky; I did not have any symptoms, just PCR testing.…It felt like I was starting over again. This was 10 years ago.” – Karen
When CML progresses, doctors will perform another round of tests, including biopsies, blood work, and mutation testing, to learn about the nature and extent of the disease.9 To better understand how and when CML becomes resistant, it is important to know how CML is monitored—what doctors test, how often, and what they are looking for.
Patients with CML who take a TKI should see their healthcare team on a routine basis to make sure the treatment is working. During the first year of treatment, these tests are generally done every 3 months. Ideally, blood tests will show that a patient with CML who is taking a TKI has achieved what is known as a “complete hematologic response” to treatment. When this happens, the patient has no symptoms; has healthy levels of WBCs and platelets; has no signs of abnormal blood cells, such as blasts in the blood; and has a normal-sized spleen, such that it cannot be felt on physical exam.9
Some patients with CML who are treated with a TKI achieve a “partial response,” which means that blood counts are better but still abnormal; there may be some blasts in the blood, and the spleen may still be enlarged. For these patients, symptoms and blood test results have improved since treatment began, but there is still room for improvement.9
If a person with Ph+ CML shows a complete hematologic response, his or her doctor then looks for a cytogenetic response with genetic testing. Results include the following9:
- “Complete cytogenetic response”; no Ph+ cells
- “Partial cytogenetic response”; between 1% and 35% of the cells are still Ph+
- “Minor cytogenetic response”; more than 35% of the cells are still Ph+.
Doctors who treat CML also use a test called polymerase chain reaction (PCR) to look at “molecular response.” This powerful blood test finds the BCR-ABL fusion genes in the patient’s body (Figure 5).20,21 When only a few cells with the BCR-ABL fusion gene are found in the bone marrow or peripheral blood (more than 1,000 times fewer than when diagnosed), the patient has achieved a “major molecular response.” “Complete molecular response” occurs when no cells with the BCR-ABL fusion gene are found in the bone marrow or peripheral blood.9

PCR Testing as a CML Monitoring Tool
Quantitative reverse transcriptase PCR is the most sensitive way to look for CML cells. This test is recommended every 3 months and uses a blood sample. Generally, this test can find as little as 1 CML cell among 1 million healthy blood cells. When this type of PCR test is negative, it is very likely that CML is well-controlled. On the other hand, if a person continues taking their TKI as directed and the PCR test shows a steady rise in the number of CML cells, then the current treatment may no longer be working. A switch in medications may be needed before the disease worsens.9
It is important to know that responses to TKI treatment can change over time, and that PCR changes should not cause panic. Instead of looking at one PCR test result that seems “out of line,” doctors advise patients to focus on the trend in test results over time. In general, PCR numbers should go down for people with CML who are being treated with a TKI. During the first several months of treatment, PCR levels usually go down significantly. After specific remission milestones are reached, the degree of reduction is generally much smaller and stability is often fine, especially if very deep remission was achieved. If CML does not respond completely or worsens based on PCR and other tests, patients may continue their current TKI to see whether it could help further, or they may switch to another TKI.9
When a rise in PCR is being assessed, it is important to focus on the point from which the rise in PCR level occurred. For example, a rise in PCR for a patient with CML who was in deep molecular remission means something different than the same increase for a patient who was not in molecular remission. When a concerning change is seen in PCR findings, doctors should perform other tests to learn whether their results are consistent.20
Roundtable participants have learned that changes in their disease, including symptoms and PCR test results, are frustrating and confusing, but “normal.” Over years of talking with their doctors and consulting with CML experts, these patients strongly recommend consulting CML experts at academic or specialty cancer centers with any questions about next steps. To substantiate their advice, consider the results of a 2015 study of community-based medical practices. This study looked at how often patients with CML underwent cytogenetic and molecular monitoring. Researchers found that patients with CML who were treated in the community were tested at lower rates than what was recommended by treatment guidelines. Specifically, both cytogenetic testing and molecular testing were done for only 1 in 4 (24%) of the patients with CML who were followed for at least 18 months in this retrospective, observational study (Figure 6).22

“Within the first year after diagnosis and initial treatment for CML, my test results and PCR findings felt like a roller-coaster ride to me. What made those hills just a bit smaller was knowing that there are more treatment options if the first one doesn’t work.” – Karen
“People with CML should not feel as if they are ‘failing’ if their PCR results are not perfect. The treatment fails the patient. This is why consistent and regular PCR testing is critical for monitoring CML status.” – Joannie
“At the beginning, I felt like I shouldn’t ever question my doctor. I’m not educated like they are. I felt guilty about getting a second opinion, like I was ‘cheating’ on my doctor. I know now that I should not have felt this way.” – Hans
Genetic Testing After CML Progression
One possible reason that CML may become resistant to treatment with TKIs is that the CML cells have changed or mutated over time. These mutations are known as “point mutations.” T315I is an example of a point mutation of the BCR-ABL gene that makes CML cells resistant to certain TKIs.9
Newer TKIs are effective for most of the point mutations on BCR-ABL. Before switching to a different TKI at the time of CML progression, all patients should undergo genetic mutation testing. Learning which BCR-ABL point mutations are present helps doctors and their patients choose the best TKI for that patient.23 Patients should be involved and have a voice in their healthcare decisions.
Resources for Patients with CML, Including Resistant CML
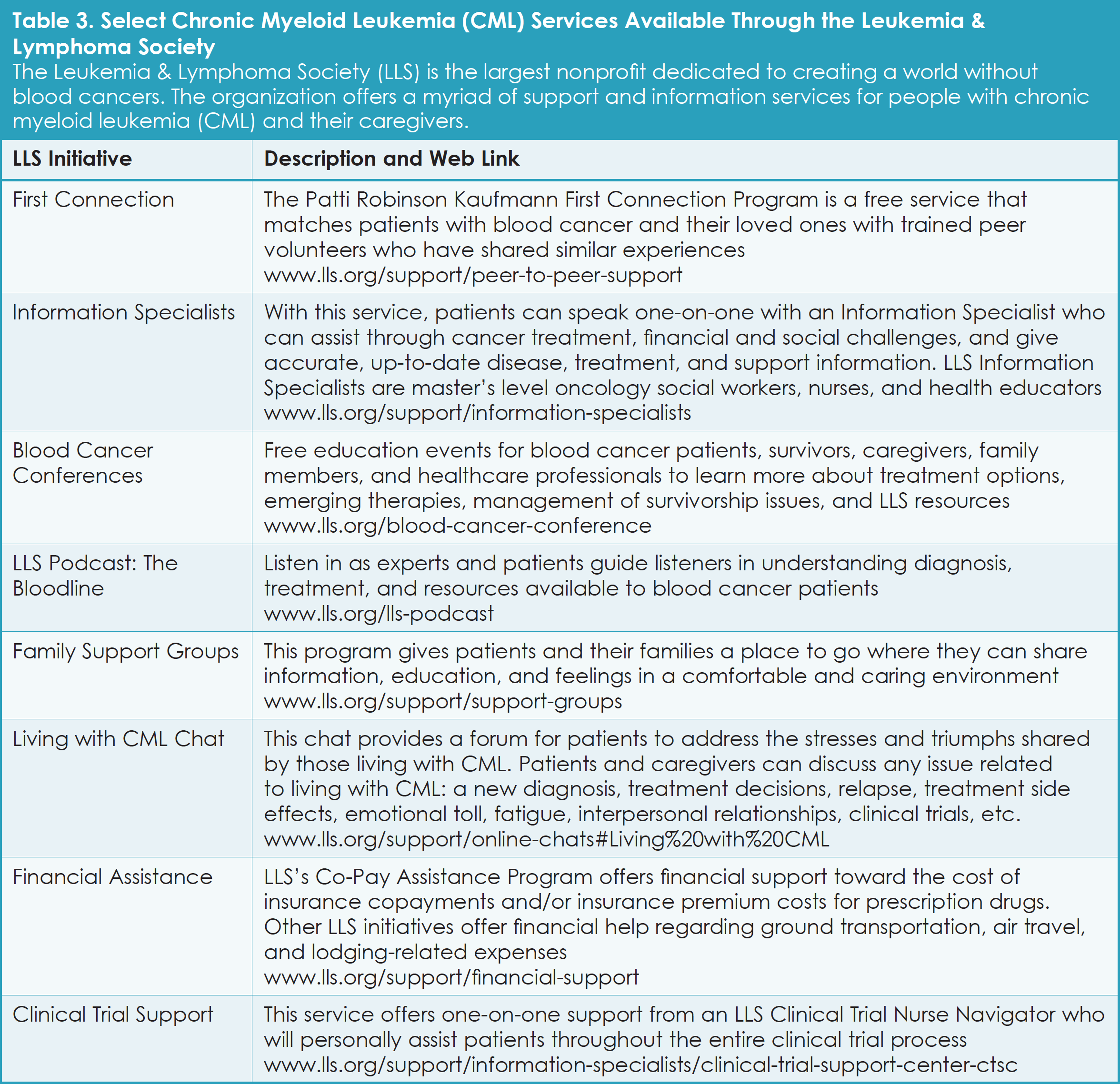
Educational resources and support are available both online and at local hospitals, libraries, and community centers. For patients with CML and their families, local and national patient organizations can be valuable sources of information and help. Table 2 provides examples of resources to consult at the time of CML diagnosis and/or if CML becomes resistant to initial treatment. Other resources provided relate to cancer-focused travel and logistics help, and clinical trials websites. Table 3 lists specific CML-focused services offered by the Leukemia & Lymphoma Society.

“Support is so important. Some patients want a safe place to say, ‘I’m tired,’ and for others to really hear them and offer support and advice.” – Hans
“Clinical trial information is really important for patients, especially if you are losing your response. I knew nothing about trials when I was diagnosed with CML. I got on a Gleevec® trial only after learning more through my doctor. If I need to switch treatments, I want to be able to learn which trials I might qualify for and be as efficient about it as I can.” – Melvin
The goals of this article were to share insights from individuals with CML who participated in the CML Roundtable meeting that was sponsored by Takeda Oncology, as well as to highlight resources that can help patients and their caregivers learn more about CML, including resistant disease. We hope that patients with CML and their caregivers can use this information to partner effectively with their medical team.
References
- Hehlmann R, Lauseker M, Saußele S, et al. Assessment of imatinib as first-line treatment of chronic myeloid leukemia: 10-year survival results of the randomized CML study IV and impact of non-CML determinants. Leukemia. 2017;31:2398-2406.
- Gleevec (imatinib) package insert. East Hanover, NJ: Novartis Pharmaceuticals; July 2018.
- Iqbal N, Iqbal N. Imatinib: a breakthrough of targeted therapy in cancer. Chemother Res Pract. 2014;2014:357027.
- Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044-1054.
- Cortes J, Saglio G, Kantarjian HM, et al. Final 5-year study results of DASISION: the dasatinib versus imatinib study in treatment-naïve chronic myeloid leukemia patients trial. J Clin Oncol. 2016;34:2333-2340.
- Brümmendorf TH, Cortes JE, de Souza CA, et al. Bosutinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukaemia: results from the 24-month follow-up of the BELA trial. Br J Haematol. 2015;168:69-81.
- Hughes TP, Saglio G, Quintás-Cardama A, et al. BCR-ABL1 mutation development during first-line treatment with dasatinib or imatinib for chronic myeloid leukemia in chronic phase. Leukemia. 2015;29:1832-1838.
- Hochhaus A, Saglio G, Larson RA, Nilotinib is associated with a reduced incidence of BCR-ABL mutations vs imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase. Blood. 2013;121:3703-3708.
- Cancer.Net. Leukemia – chronic myeloid – CML. Last revised June 19, 2018. www.cancer.net/cancer-types/leukemia-chronic-myeloid-cml. Accessed July 5, 2019.
- American Cancer Society. If you have chronic myeloid leukemia (CML): What is leukemia? www.cancer.org/cancer/chronic-myeloid-leukemia/if-you-have-chronic-myeloid-leukemia.html. Accessed July 5, 2019.
- U.S. Department of Health and Human Services Medline Plus website. BCR ABL Genetic Test. Page last updated February 4, 2019. https://medlineplus.gov/lab-tests/bcr-abl-genetic-test/. Accessed November 18, 2019.
- Cleveland Clinic. What is cancer? https://my.clevelandclinic.org/departments/cancer/patient-education/wellness-prevention/what-is-cancer. Accessed July 5, 2019.
- Cancer Treatment Centers of America. Types of leukemia. www.cancercenter.com/cancer-types/leukemia/types. Accessed July 9, 2019.
- National Cancer Institute. How imatinib transformed leukemia treatment and cancer research. Updated April 11, 2018. www.cancer.gov/research/progress/discovery/gleevec. Accessed July 9, 2019.
- Druker BJ, Sawyers CL, Kantarjian H, et al. Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med. 2001;344:1038-1042.
- American Cancer Society. Targeted therapies for chronic myeloid leukemia. Last revised June 19, 2018. www.cancer.org/cancer/chronic-myeloid-leukemia/treating/targeted-therapies.html. Accessed July 9, 2019.
- Leukemia & Lymphoma Society. Treatment outcomes. www.lls.org/leukemia/chronic-myeloid-leukemia/treatment/treatment-outcomes. Accessed July 9, 2019.
- Kropf P, Barnes G, Tang B, Pathak A, Issa J-P. Burden of tyrosine kinase inhibitor failure in patients with chronic myeloid leukemia. J Leuk. 2015;3:1.
- de Lavallade H, Apperley JF, Khorashad JS, et al. Imatinib for newly diagnosed patients with chronic myeloid leukemia: incidence of sustained responses in an intention-to-treat analysis. J Clin Oncol. 2008;26:3358-3363.
- MyPCR.org. My PCR Frequently Asked Questions. September 2012. http://mypcr.org/wp-content/uploads/2019/02/MY-PCR_FAQ_English.pdf. Accessed July 13, 2019.
- Mahon FX1, Etienne G. Deep molecular response in chronic myeloid leukemia: the new goal of therapy? Clin Cancer Res. 2014;20:310-322.
- Di Bella NJ, Bhowmik D, Bhor M, et al. The effectiveness of tyrosine kinase inhibitors and molecular monitoring patterns in newly diagnosed patients with chronic myeloid leukemia in the community setting. Clin Lymphoma Myeloma Leuk. 2015;15:599-605.
- Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring. Am J Hematol. 2018;93:442-459.
USO-NON-0018 05/20











