When people think of cancer, they often think of diseases such as breast or lung cancer. But approximately 1 in 5 people living with cancer in the United States have been diagnosed with a rare cancer, each type affecting less than 20,000 Americans a year.1 Although some rare cancer types, such as cancer of the bile duct called cholangiocarcinoma (CCA), may be unknown to most people, rare cancers also include subsets of even the most well-known cancers. In addition, all pediatric cancers are considered rare.2
As with many rare disorders, the infrequency of these cancers and lack of education and awareness about them, have historically made the research and treatment difficult. In fact, the 5-year survival rate is lower for patients with rare cancer than for those diagnosed with a more common cancer.1
The good news is that advancements in cancer care, including insights from genomic research about gene alterations in cancer, help address this disparity.
Genomic Mutations
In recent decades, research has shown that cancer is caused by changes, or mutations, to genes that control how cells function. This has helped to change our understanding of cancer from a disease characterized mainly by its location in the body (such as lung or breast) to a disease of our DNA, also known as the “genome.”
With this understanding, researchers have been able to develop therapies to target the underlying gene mutations that cause cancer. These targeted therapies are often less toxic and more effective than older, less precise treatments, such as chemotherapy.
Genomic Testing
Genomic profiling or testing (also known as biomarker testing) looks for alterations and mutations in the DNA of a patient’s cancer by testing samples of the tumor tissue or blood.
Genomic testing is different from genetic testing. Genomic testing looks for DNA mutations that are not inherited from a parent or grandparent and are unique to cancer cells, whereas genetic testing looks for mutations that are inherited from a family member and are found in every cell in the body.
Information from a genomic test can potentially be used to match patients with a therapy that is appropriate for that unique cancer, which is called “precision medicine,” and includes FDA-approved targeted therapies or immunotherapies. Such testing can also help to identify appropriate clinical trials for a patient.
The use of genomic testing has created vast amounts of data on the various mutations that cause cancer. These data about genomic information are helping to accelerate cancer research, leading to advances in treatments for patients with rare cancers.
Precision Medicine for Rare Cancers
Here are a few examples demonstrating how genomic research has supported the oncology community’s commitment to advancing precision medicine for rare cancers.
CCA is a rare cancer of the bile duct (the thin tubes connecting the liver to the small intestine). About 8,000 Americans each year are diagnosed with CCA, which is less than 0.5% of all cancers.3 For many years, there was no FDA-approved targeted treatment for people with this type of cancer—only chemotherapy, radiation, and surgery.3 The diagnosis of CCA often happens in the late stages of the disease, which can limit the available treatment options and lead to a poor prognosis for the patient.
However, insights gathered from genomic testing data reveal, for example, that 10% to 16% of patients with CCA have a specific mutation that is driving the growth of that cancer.4 Scientists used this information to develop targeted treatments that block these mutations—known as FGFR2 gene fusion or other rearrangement—thereby preventing the cancer from growing and spreading.
New Therapies for Rare Mutations
Recently, the FDA approved new treatments for patients with CCA whose tumor has an FGFR2 gene fusion, as well as a new treatment for patients with CCA and an IDH1 mutation. Genomic testing makes it possible to determine which patients have either alteration in their cancer, which can guide the choice of the targeted therapy for the individual patient.
Genomic research is also enabling scientists to develop targeted therapies that can help patients with cancer, regardless of where the tumor is located. These so-called pan-tumor treatments are prescribed based on the genomic alterations in the tumor, which are allowing researchers to bring new treatments to many rare cancers faster than before.
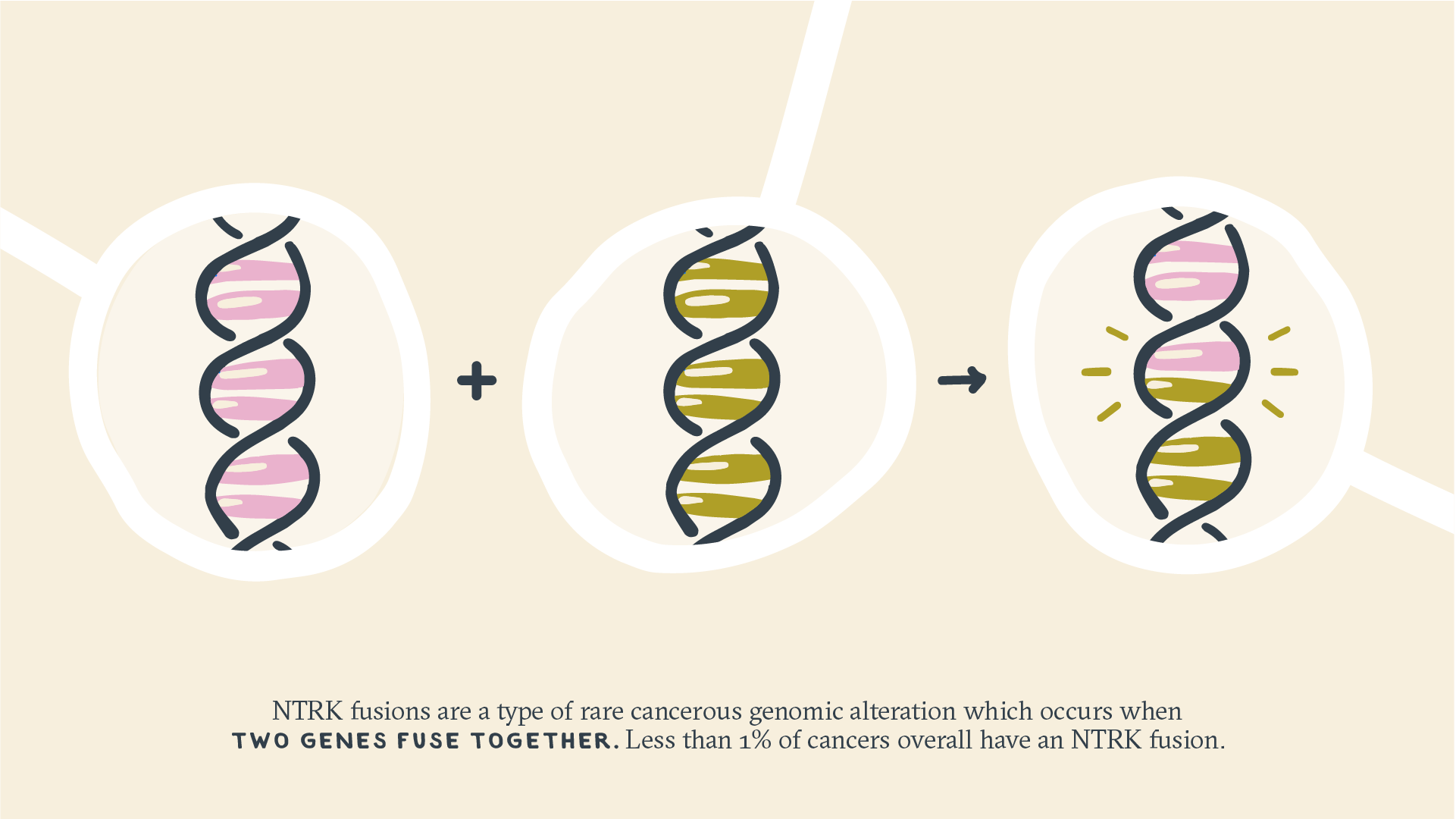
Neurotrophic receptor tyrosine kinase (NTRK) fusions are another type of rare malignant genomic alteration that occurs when 2 genes fuse together. These changes can cause normal cells to become cancerous anywhere in the body, including the breast, thyroid, and colon.5
Less than 1% of cancers overall have an NTRK fusion, but the prevalence can vary greatly by cancer type. In several rare cancers, such as secretory breast cancer and infantile fibrosarcomas, more than 90% of patients have an NTRK fusion. But more common cancers, such as colorectal cancer and invasive breast cancer, rarely have an NTRK fusion (less than 1% of cases).6,7
A tissue-based or blood-based genomic test makes it possible to identify patients whose cancer has an NTRK fusion. For those patients, FDA-approved therapies that target NTRK fusions may offer important treatment options, regardless of the cancer’s location.
Genomic information can also reveal critical insights beyond the specific gene mutation. For example, using broad genomic testing that looks at a large number of genes, researchers identified a genomic signature called tumor mutational burden (or TMB) that measures the overall number of mutations in a patient’s cancer rather than in a specific mutation.
Using an FDA-approved genomic test to determine the level of TMB, doctors are now able to prescribe an FDA-approved immunotherapy that treats cancer by harnessing the power of the immune system, to patients with high TMB levels, regardless of their tumor type or location. For some patients with a rare cancer, the discovery of this actionable genomic signature has opened a door to this innovative treatment that may otherwise have remained closed.
Genomic Testing Is Key
As these examples show, genomic research has yielded profound changes to the treatment of rare cancers.
A critical driver of this progress is genomic testing, which gives doctors information to help their patients with rare cancer get the best treatments today, and contributes to the growing body of data being used to drive forward advancements in research for tomorrow.
To ensure access to the best treatment for patients with rare cancer now, as well as the accelerated development of future treatment options, it is imperative that all patients with cancer have access to genomic testing.
References
- DeSantis CE, Kramer JL, Jemal A. The burden of rare cancers in the United States. CA: A Cancer Journal for Clinicians. 2017;67(4):261-272.
- National Cancer Institute. Rare cancers of childhood treatment (PDQ)–health professional version. Updated February 10, 2021. www.cancer.gov/types/childhood-cancers/hp/rare-childhood-cancers-pdq.
- American Cancer Society. Key statistics for bile duct cancer. Updated July 3, 2018. www.cancer.org/cancer/bile-duct-cancer/about/key-statistics.html.
- Pellino A, Loupakis F, Cadamuro M, et al. Precision medicine in cholangiocarcinoma. Translational Gastroenterology and Hepatology. 2018;3(7):40.
- NTRKers. NTRK gene fusion cancer. https://ntrkers.org/ntrk-fusion-cancer/.
- Forsythe A, Zhang W, Strauss UP, et al. A systematic review and meta-analysis of neurotrophic tyrosine receptor kinase gene fusion frequencies in solid tumors. Therapeutic Advances in Medical Oncology. 2020;12:1-10.
- Penault-Lorca F, Rudzinski ER, Sepulveda AR. Testing algorithm for identification of patients TRK fusion cancer. Journal of Clinical Pathology. 201;72(7):460-467.