Chronic lymphocytic leukemia, or CLL, is the most common type of leukemia in adults. According to the American Cancer Society, approximately 20,940 people will be diagnosed with CLL in 2018, and 4,510 will die from the disease. CLL develops when the bone marrow produces too many white blood cells, called lymphocytes.
Because CLL is generally a slow-growing disease, patients with CLL may not have any symptoms until later, when the disease progresses. The most common signs of CLL include swollen lymph nodes, tiredness, weight loss, and fever.
The outlook (prognosis) for patients with CLL depends on several factors, including the patient’s overall health, the stage of CLL, whether the cancer responds to treatment or comes back, and whether the patient has certain genet-ic abnormalities or mutations (changes).
Knowing what type of genetic changes or mutations you have is becoming increasingly important, because:
- Some DNA abnormalities or mutations are associated with poor prognosis (or outcomes) and others are associated with good outcomes
- Some of these genetic changes respond better to some treatments, which can help to determine the best treatment
- Clinical trials are investigating the best treatment for some mutations in CLL, so a doctor can recommend a clinical trial for a patient with a specific mutation.
Therefore, knowing if your CLL has any of these genetic abnormalities can help your doctor to select the best treatment for you.
Meeting Your Genetic Mutations
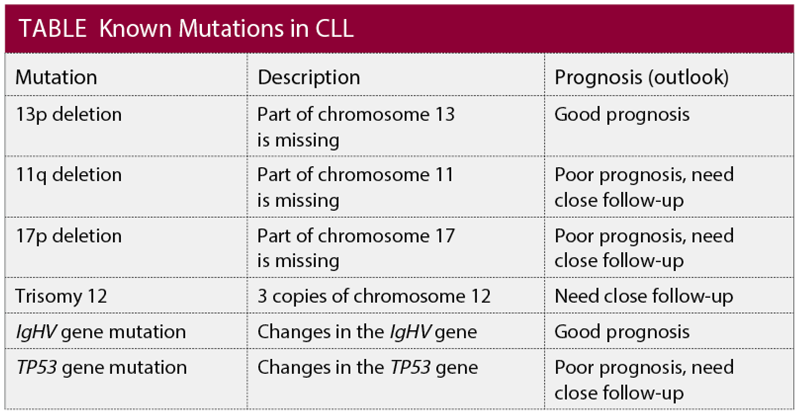
Mutations or changes in the genes or DNA can come from changes in our genes or from other changes in our DNA. Sometimes, part of a chromosome is missing; these are called “deletions.” In other mutations, there are extra copies of chromosomes. In CLL, several genetic changes can help to determine the prognosis of the patient and help to decide on the best treatment.
A common abnormality in CLL is a deletion in the 17 chromosome, which is called “17p deletion.” Less common deletions in other chromosomes are “13p deletion” and “11q deletion.”
Other known mutations in CLL are “trisomy 12,” which has 3 copies of chromosome 12, and the 2 gene mutations—TP53 and IgHV gene mutations.
Overall, having the 13p deletion or the IgHV gene mutation is good, because these mutations are associated with better prognosis; they improve the chances for the patient to have good outcomes.
By contrast, having the 17p deletion, 11q deletion, or the TP53 gene mutation, trisomy 12, or no IgHV mutation is associated with poor prognosis for the patient (see Table).
Why Should You Have Genetic Testing?
Genetic testing can help doctors discover whether your CLL has any genetic abnormality or mutation. Several genetic tests are available to detect mutations related to cancer. These tests usually use the patient’s own blood to identify any changes in the DNA.
Fluorescence in situ hybridization (or FISH) is a common genetic test that uses fluorescent probes to detect if parts of the DNA are missing. Karyotyping is another type of genetic test used in CLL. It looks at all the chromosomes in our body and can find whether parts of any chromosome are missing, or if the person has extra chromosomes.
Testing for mutations is important at diagnosis, as well as throughout the disease. According to the Leukemia & Lymphoma Society, “It is important to have another FISH test prior to additional treatments because this test can help your doctor determine the next therapy. New mutations can develop over time or as a result of past treatments.”1
And according to hematologist/oncologist Dr. Danielle M. Brander, less than 10% of patients with CLL have the 17p deletion or TP53 gene mutation when they are first diagnosed, but this number can jump to 50% after the first treatment, or when the disease doesn’t respond to chemotherapy.2
Genetic Testing and CLL Treatment
Genetic tests are becoming more and more important, because different treatments can target specific mutations in CLL (and in other cancers), which helps doctors to personalize the patient’s treatment.
A new article emphasizes the importance of genetic testing in CLL: “At the time of treatment decision-making, genetic and molecular data are critical to select the most appropriate therapy for an individual patient and can be useful in identifying patients as candidates for clinical trials targeting genetically unfavorable-risk disease.”3
For a long time, the treatment for CLL was limited mainly to chemotherapy. Although chemotherapy works well in some patients, not all patients with CLL respond to chemotherapy.
Patients with the IgHV gene mutation usually benefit from chemotherapy, but patients without a mutation in the IgHV gene, or patients with 17p deletion or the TP53 gene mutation don’t respond to chemotherapy. However, newer therapies are available for those patients. And because chemotherapy can have serious side effects, understanding which mutations you have can help avoid these unnecessary side effects and get the patient the most appropriate treatment faster.
In the past 4 years, newer drugs known as “targeted therapies” have changed how CLL is being treated. These drugs give patients who normally would not respond to chemotherapy, based on their mutations, more treatment options. Some of these drugs specifically indicate which mutations patients should have to benefit most from treatment.
For example, Imbruvica (ibrutinib) is approved by the FDA as the first treatment for patients with CLL and 17p deletion. Another treatment for patients with CLL and 17p deletion is Venclexta (venetoclax), which is approved by the FDA for use after other treatments for patients with CLL with or without 17p deletion.
Other recent treatments approved for CLL but not for specific mutations are Zydelig (idelalisib), in combination with Rituxan (rituximab), for patients with relapsed (returning) or refractory CLL, and the newest treatment, Copiktra (duvelisib), which was approved by the FDA for relapsed or refractory CLL in September 2018.
Ask Your Doctor About Genetic Testing
CLL treatment guidelines recommend that all patients with CLL undergo genetic testing, but many patients do not get tested. Treatment for CLL is quickly changing, and doctors are focused on tailoring treatment so that patients can benefit most from therapy. This is why it is important now, more than ever before, to speak with your doctor about getting tested.
Key Points
- CLL is the most common type of leukemia in adults
- Because CLL grows slowly, patients may not have any symptoms until the disease progresses
- Genetic testing can help doctors discover whether your CLL has any mutations
- In the past 4 years, newer drugs known as “targeted therapies” have changed how CLL is treated
References
1. Leukemia & Lymphoma Society. The CLL Guide: Information for Patients and Caregivers: Chronic Lymphocytic Leu-kemia. Revised 2017. www.lls.org/sites/default/files/National/USA/Pdf/Publications/PS48_CLL_Guide_8_17FINAL.pdf.
2. Brander D. CLL prognosis and treatment options. Targeted Oncology. January 12, 2018. www.targetedonc.com/case-based-peer-perspectives/chronic-lymphocytic-leukemia/brander-relapsed-cll/cll-prognosis-and-treatment-options.
3. Mato A, Nabhan C, Kay NE, et al. Prognostic testing patterns and outcomes of chronic lymphocytic leukemia patients stratified by fluorescence in situ hybridization/cytogenetics: a real-world clinical experience in the Connect CLL Registry. Clinical Lymphoma, Myeloma & Leukemia. 2018;18(2):114.e2-124.e2.
Patient Resources
American Cancer Society
www.cancer.org/cancer/chronic-lymphocytic-leukemia/treating/targeted-therapy.html
CLL Society
https://cllsociety.org
Leukemia & Lymphoma Society
www.lls.org/sites/default/files/file_assets/cllguide.pdf













