TLG1784-2
Non–small-cell lung cancer (or NSCLC) is the most common form of lung cancer, accounting for about 80% to 85% of all lung cancer cases in the United States.1 Given the complexity of cancer, the rapid development of new treatments, and the large amount of information available, it may be challenging for patients to find materials to help them better understand their disease.
The goal of this 4-part series of articles is to present brief overviews of concepts that may be important to caregivers and patients with NSCLC. In this second part, we present an overview of the current treatment landscape for NSCLC, including a brief description of the most often used treatments, as well as a list of questions that patients can ask their treatment team.
Treating Patients With Non–Small-Cell Lung Cancer
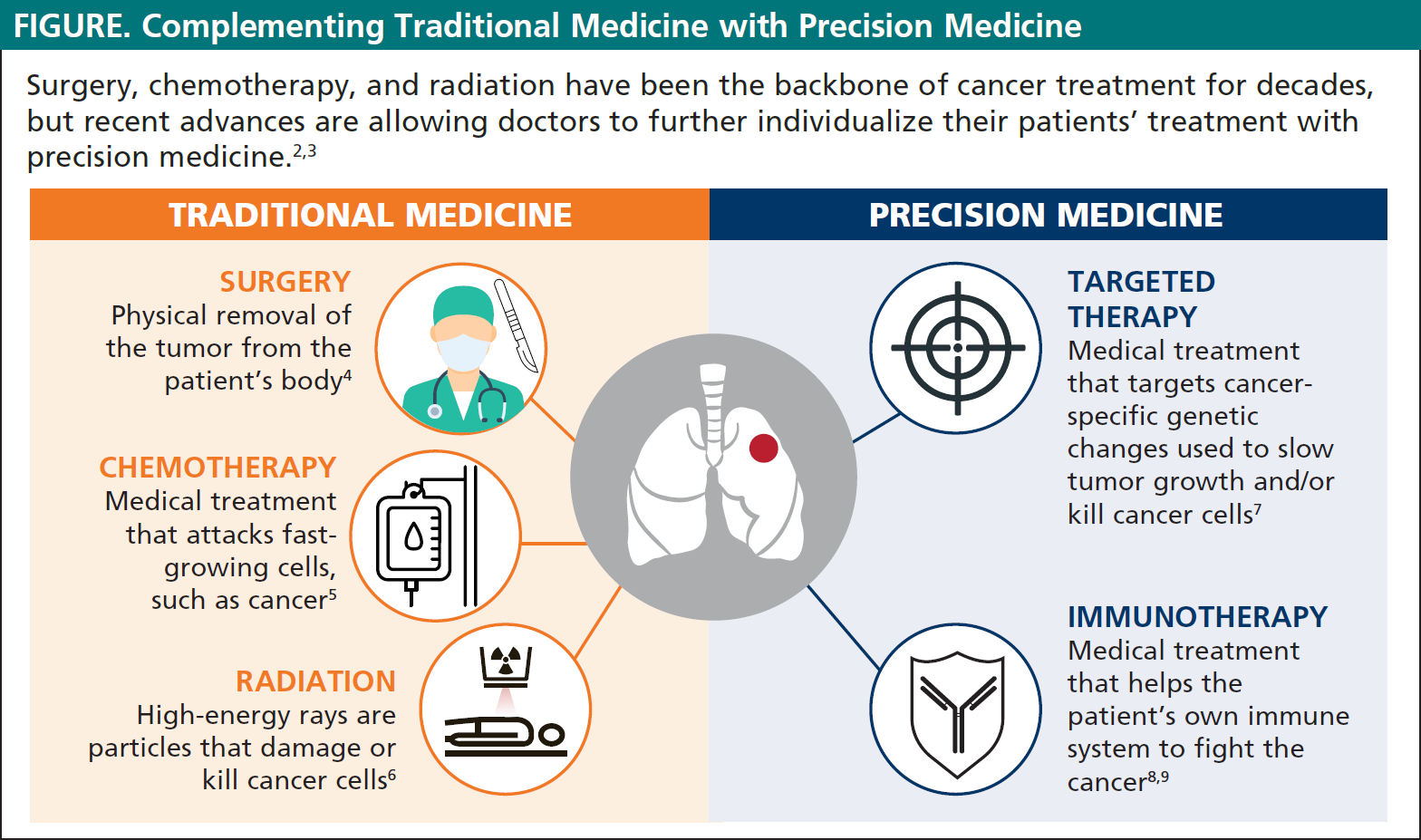
Cancer treatment has changed significantly in recent years. Many different therapies are available today for patients with NSCLC (see Figure below). Doctors no longer treat each patient with NSCLC in the same way; instead, physicians develop personalized treatment plans for each patient based on factors such as the stage of cancer, the cancer’s unique characteristics, and the patient’s overall health status.
One key development in modern cancer treatment has been the discovery of cancer-specific genetic changes or mutations (also known as biomarkers) that can be directly targeted by medications known as targeted medications.2 These targeted therapies are the foundation of precision medicine, an approach to patient care that allows doctors to select treatments that are most likely to help individual patients based on the genetic characteristic of their disease.2,3
Below, we briefly review the types of therapies that patients with NSCLC may receive. We also explain the importance of clinical trials in developing new medicines and what patients should expect if they choose to participate in a clinical trial.
Overview Of Therapies For Non–Small-Cell Lung Cancer
Surgery
Surgery is a type of procedure in which the cancer is physically removed from the body by a surgeon.4
Surgery works best for solid tumors that are contained in one area of the body. In fact, most NSCLC tumors that are at stage I or stage II disease are removed by surgery.8 However, if the cancer has spread (metastasized), surgery may not be the best treatment.10 Patients may also receive neoadjuvant therapy before surgery, or adjuvant therapy after surgery (see Box below).8
What Are Neoadjuvant and Adjuvant Therapy
Surgery alone may not be sufficient to treat a patient's cancer. Sometimes the treatment plan may include presurgical treatment (called neoadjuvant therapy) or follow-up treatment (called adjuvant therapy) after surgery.
Neoadjuvant therapy is treatment given before a main treatment. Neoadjuvant therapy may include chemotherapy or radiation therapy before surgery, used to shrink the tumor and make it easier to remove.
Adjuvant therapy is additional cancer treatment given after the primary treatment to lower the risk that the cancer will come back (relapse). Adjuvant therapy may include chemotherapy, radiation therapy, targeted therapy, or immunotherapy.
Chemotherapy
Chemotherapy is a medical treatment that attacks fast-growing cells, such as cancer.5 Depending on the characteristics of the cancer, including its stage and whether it has spread, different chemotherapy drugs may be recommended.5
For patients with stage I or stage II lung cancer, a combination of 2 chemotherapy drugs is often used as treatment.8 Patients with stage III or stage IV lung cancer may receive chemotherapy alone or in combination with other treatments, depending on their overall health.8 A targeted therapy, which will be discussed later, may also be added to chemotherapy in patients with stage III or stage IV lung cancer.
Radiation
Radiation uses high-energy rays or particles to kill cancer cells. The 2 types of radiation available are6:
- External beam radiation therapy: A machine that acts like a very strong x-ray aims radiation directly at the cancer.
- Brachytherapy: A small amount of radioactive material is placed inside the body close to the cancer. Radiation may be used as the main treatment (or sometimes along with chemotherapy) in cases when the tumor in the lung cannot be removed because of its size or location, when the patient is not healthy enough for surgery, or if the patient does not want surgery.6,8
Radiation Therapy: Photons versus Protons
Traditionally, radiation therapy has been delivered using photons, which are energetic particles of light (better known as x-rays) that can penetrate tissues and kill cancer cells. Although photons can pass through healthy tissue, they often damage healthy cells along the way, which may lead to side effects.11,12 For example, one common side effect of photon radiation therapy is a skin rash that looks like a sunburn.11,12
Like photon therapy, protons, which are heavy particles at the center of atoms, can also penetrate tissues and kill cancer cells. However, unlike photons, the location where protons release their energy can be better controlled by radiation oncologists.13 By controlling the depth to which the energy is released, radiation oncologists can target the tumor itself, while minimizing damage to healthy tissues.13
The relative benefits of protons compared with photons is a source of debate and scientific research. As of this writing, proton therapy is currently available at 32 centers throughout the United States and 3 additional sites are being built or developed.14
Targeted Therapy
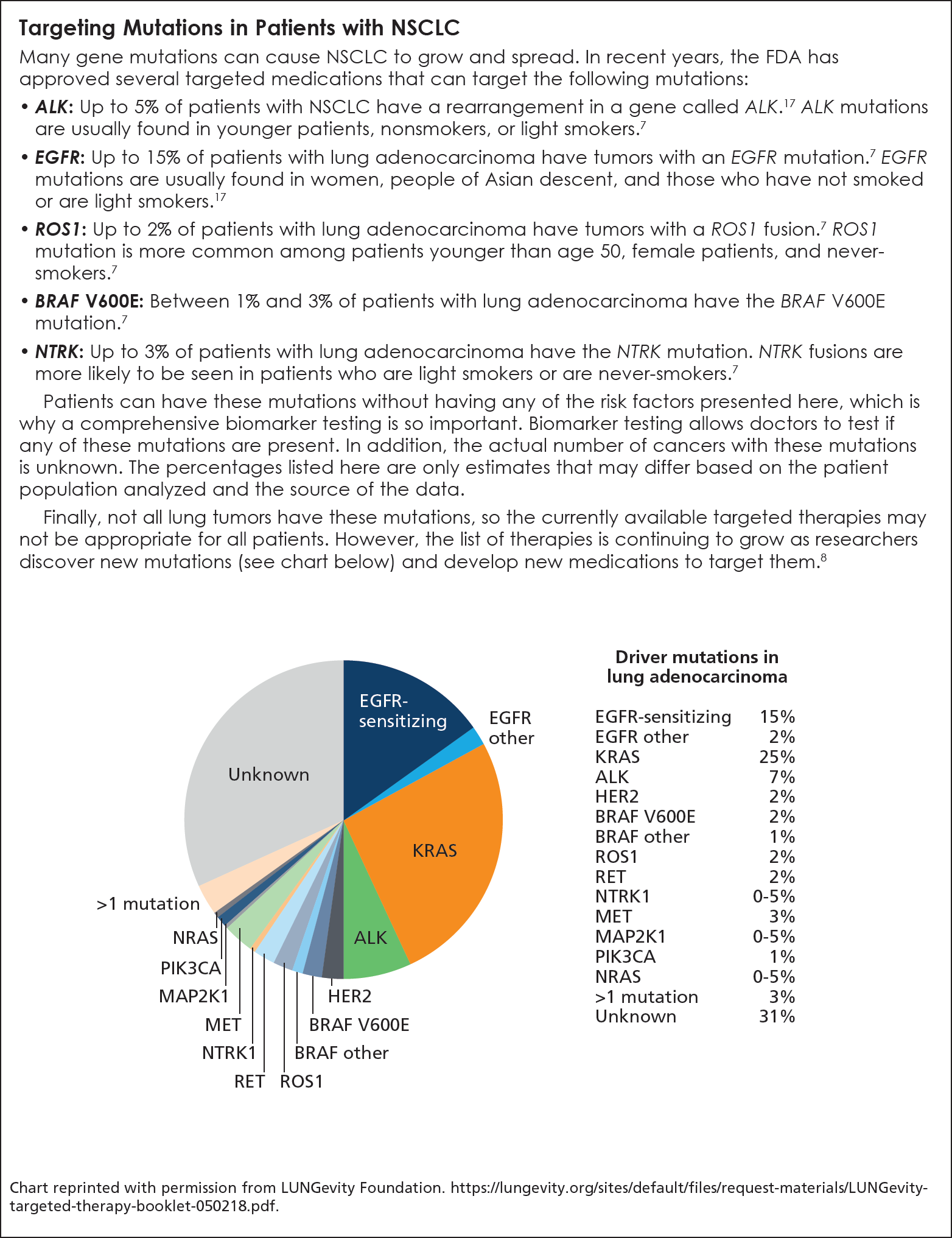
In patients with NSCLC, many genetic changes (or mutations) have been identified that enhance the tumor’s ability to grow and spread (see Box). Targeted therapy is a form of medical treatment that focuses on the genetic changes that are specific to cancer cells and can be used to slow their growth and/or kill them.15 These genetic changes are specific to each person’s cancer, and therefore they cannot be passed to a patient’s children; they may not even be present in all patients with the same type of cancer.
Targeted therapies are used in the treatment of patients with stage III or stage IV cancer.16 Because targeted therapies only work in patients whose tumors have that specific targeted mutation, the cancer must first be biopsied and tests must be performed to confirm that the patient has that specific mutation.
Immunotherapy
Immunotherapy is a medical treatment that helps the body’s own immune system to fight cancer. 8,9 Some types of immunotherapy are also called biologic therapies. Immunotherapy is relatively new and has emerged as a novel treatment option for certain types of lung cancers.
Immunotherapy includes treatments that work in different ways. They can boost the patient’s own immune system in a very general way, or they can train the immune system to attack specific cancer cells.8
Today, immunotherapy is often the first treatment used for patients with stage III or stage IV NSCLC, and can be given with or without chemotherapy.8 Immunotherapy can also be used for patients whose cancer has started to grow again after initial treatment with chemotherapy or other drugs.
Future Therapies
As a result of the efforts of researchers, healthcare professionals, and patients who participate in clinical trials, new therapies for NSCLC are continuing to be investigated. Novel scientific treatment approaches and emerging areas of interest include:
- Additional targeted therapies
- New immunotherapies
- Angiogenesis inhibitors, which can prevent the formation of new blood vessels that cancers need to grow
- Immunomodulators, which can enhance or dampen the patient’s immune response
- Exploration of the tumor microenvironment, which can help us understand how the local environment around the tumor, including the surrounding blood vessels, cells, and signaling molecules, can be used to treat the cancer
The Importance of Clinical Trials
All cancer treatments must first be evaluated in a clinical trial. Clinical trials are research studies that allow doctors to find new ways to discover and improve treatments for people with diseases, including cancer.18 Clinical trials are the final step in a long process that begins with research in a laboratory. Before any new cancer treatment is tested on people, researchers work for many years to understand the drug’s effects, including any potential side effects, on cancer cells in a laboratory and in animals.18
Clinical trials determine if new treatments are safe and effective, and whether they work better than the currently available treatments. Participation in a clinical trial is completely voluntary, and patients are free to leave the study at any time, without needing to give any explanations. In fact, all patients must be informed about the benefits and risks (see Box below) of participating in a clinical trial before providing their consent to participate, and they can revoke their consent at any time, for any reason.
Clinical Trials
Possible benefits of participating in a clinical trial
- Patients will have access to a new treatment that is not available outside of the trial.
- Patients will be closely monitored, oftentimes having more medical attention than normal.
- If the treatment being studied is more effective than the standard treatment, study participants may be among the first to benefit.
- The trial may help scientists learn more about cancer and help people in the future.
Possible risks of participating in a clinical trial
- The new treatment may not be better than the treatment that is currently available.
- New treatments may have side effects that doctors do not expect or that are worse than those of the standard treatment.
- Patients may be required to make more visits to the doctor than if they were receiving standard treatment.
When considering the benefits and risks of clinical trials, it is also important to understand the role of placebo in clinical trials (see Box below).
Placebo or Standard of Care
Whether a pill or an injection, a placebo is designed to look like the actual medicine being tested, but it is not active. Placebos are used to prevent patients and their doctors from knowing whether they are receiving the actual treatment, which could affect the results of a clinical study. Placebos are rarely used in cancer treatment clinical trials. They may be used when there is no standard treatment, or when researchers want to study combinations of treatments. When placebos are used, patients usually receive them alongside standard-of-care therapy (see next paragraph).
Standard-of-care therapy is accepted by medical experts as a proper treatment for that disease. In other words, it is the treatment that patients would have received if they had not joined the trial. Participants in a clinical trial will always be told if the study uses a placebo or standard of care.
What Questions Can Patients Ask Their Doctor?
- Which treatments are appropriate for my stage of cancer?
- Targeted therapy, immunotherapy, chemotherapy, combination therapy, local treatment—how do I know which therapy is appropriate for me?
- Will any tests be needed before making a treatment decision?
- How will the doctor know if the treatment is working?
- What are the possible side effects or risks of treatment?
- Is there anything that can be done to help control my symptoms or to treat my side effects?
- Are clinical trials an option for me? Are they covered by my insurance?
- What is the purpose of the trial?
- How long will I be in the trial?
- What kinds of tests and treatments are involved?
- If I begin the trial and receive a placebo, will I be able to switch to the study drug at some point?
- What are the consequences if I no longer want to participate in the trial?
- How will I be told about the trial’s results?
- Who can I speak with about questions I have during and after the trial?
- Is there someone I can talk to who has been in the trial?
- How do the possible risks and benefits of this trial compare with those of the standard treatment?
References
- Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clinics in Chest Medicine. 2011;32:605-644.
- Pennell NA, Arcila ME, Gandara DR, West H. Biomarker testing for patients with advanced non-small cell lung cancer: real-world issues and tough choices. American Society for Clinical Oncology Education Book. Vol 39. 2019:531-542.
- National Cancer Institute. Precision medicine in cancer treatment. www.cancer.gov/about-cancer/treatment/types/precision-medicine. October 3, 2017.
- National Cancer Institute. Surgery to treat cancer. www.cancer.gov/about-cancer/treatment/types/surgery.
- American Cancer Society. Chemotherapy for non-small cell lung cancer. www.cancer.org/cancer/lung-cancer/treating-non-small-cell/chemotherapy.html. October 1, 2019.
- National Cancer Institute. Radiation therapy to treat cancer. www.cancer.gov/about-cancer/treatment/types/radiation-therapy#HRTWAC. January 8, 2019.
- LUNGevity. Targeted therapy. https://lungevity.org/for-patients-caregivers/lung-cancer-101/treatment-options/targeted-therapy.
- Hirsch FR, Scagliotti GV, Mulshine JL, et al. Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389(10066):299-311.
- American Cancer Society. How immunotherapy is used to treat cancer. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/ what-is-immunotherapy. December 27, 2019.
- Cancer Research UK. About surgery. www.cancerresearchuk.org/about-cancer/cancer-in-general/treatment/surgery/about. April 12, 2016.
- LUNGevity. Radiation therapy. lungevity.org/for-patients-caregivers/lungcancer-101/treatment-options/radiation-therapy. August 6, 2019.
- American Society of Clinical Oncology. Side effects of radiation therapy. www.cancer.net/navigating-cancer-care/how-cancer-treated/radiation-therapy/sideeffects-radiation-therapy.
- Brooks ED, Ning MS, Verma V, et al. Proton therapy for non-small cell lung cancer: the road ahead. Translational Lung Cancer Research. 2019;8(suppl 2):S202-S212.
- National Association for Proton Therapy. Proton therapy centers in the U.S. www.proton-therapy.org/map/.
- American Cancer Society. How targeted therapies are used to treat cancer. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/targeted-therapy/what-is.html.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Non-Small Cell Lung Cancer. Version 1.2020. www.nccn.org/professionals/physician_gls/pdf/nscl.pdf.
- American Cancer Society. Targeted therapy for non-small cell lung cancer. www.cancer.org/content/cancer/en/cancer/lung-cancer/treating-non-small-cell/targetedtherapies.html.
- National Cancer Institute. Clinical trials information for patients and caregivers. www.cancer.gov/about-cancer/treatment/clinical-trials.
Articles in this Series
- Staging of Non–Small-Cell Lung Cancer: A Guide for Patients
- Treatment of Non–Small-Cell Lung Cancer: A Guide for Patients
- Biomarker Testing in Non–Small-Cell Lung Cancer: A Guide for Patients
- The Patient Journey in Non–Small-Cell Lung Cancer: An Overview