Immune cells in the body can usually detect the changes in cancer cells, both genetic changes and changes to the cell’s appearance. Generally, the immune system can activate its normal functioning to help kill off cancer cells. But cancer cells themselves can change the immune cell activity and use it to prevent recognition and destruction by the immune system and then grow without being killed off.1 Immunotherapy can mark the cancer cells so the immune system can easily find and destroy the cancer cells in the body. Immunotherapy can also boost the immune system’s effectiveness in fighting cancer.2
What Is Immunotherapy?
There is a lot of media attention around immunotherapy for treatment of cancer. Immunotherapy is not just 1 type of treatment; it is a variety of treatments that all use the immune system in different ways to help fight cancer. Immunotherapy falls into a category of cancer therapy called biologic therapy and uses the immune system, or something made by the immune system, to fight off cancer cells.
The goal of immunotherapy is to get your own immune system to attack and destroy cancer cells. Initially, immunotherapy focused on increasing the immune system’s antitumor response by targeting specific molecules. There are some cancers in which immunotherapy works very well, and others in which it does not work as well.
The first types of immunotherapy used cytokines (specific substances secreted by immune cells) and similar substances manufactured to treat cancer, including the substances interleukin-2 (IL-2) and interferon (IFN) alfa-2b. These were both used to treat melanoma and renal cell carcinoma. Unfortunately, the early immunotherapy was not very effective and had significant side effects.3
In the past 8 to 10 years, advances have been made in immunotherapy that now provide treatments for cancers such as head and neck, bladder, kidney, gastric, ovarian, liver, and lung (both non–small cell and small cell), as well as melanoma, Hodgkin lymphoma, Merkel cell carcinoma, and some types of leukemia. These treatments are being approved for other cancers all the time.4
Types of Immunotherapy
Immune Checkpoint Inhibitors
Checkpoint proteins (PD-1) work to regulate the response of the immune system to cancer cells; they can be turned off to keep the T-cells (a type of white blood cell) from attacking other cells in your body.5 In cancer; the cancer cells find ways to avoid the immune system altogether, thus growing without stopping. Immune checkpoint inhibitors (ICIs) are drugs that increase the response of the immune system to a tumor. When PD-1 attaches to the protein PD-L1 on cells, it tells them to leave the other cells alone. When you block PD-1 or PD-L1, it will allow T-cells to kill cancer cells.5 They “release the brakes” and allow the immune system to respond to the cancer cell “invasion.”1 Another target of ICIs is CTLA-4, which is another protein on T-cells that can be used as a switch to keep the immune system in check.5 ICIs do not actually kill cancer cells, but they interfere with the ability of cancer cells to avoid the immune system efforts and allow the immune system to kill off more cancer cells.2
Drugs in the PD-1 inhibitor class include pembrolizumab (Keytruda), nivolumab (Opdivo), and cemiplimab (Libtayo). These drugs are used to treat melanoma, non–small cell lung cancer, kidney cancer, bladder cancer, head and neck cancer, and Hodgkin lymphoma.5 They are being used in clinical trials for many other types of cancer as well.
PD-L1 inhibitor drugs include atezolizumab (Tecentriq), avelumab (Bavencio), and durvalumab (Imfinzi). These drugs are primarily used for treatment of bladder cancer, non–small cell lung cancer, and Merkel cell carcinoma. They are also being tested in clinical trials for other types of cancer.
Drugs that target CTLA-4 include the drug ipilimumab (Yervoy). This is a monoclonal antibody that attaches to CTLA-4 and stops it from working altogether. This drug is currently used to treat melanoma.
The main problem is that these drugs can allow the immune system to attack normal cells in the body and attack organs other than those it is targeting for cancer treatment. Some people experience serious side effects because of attacks on normal organs.5 Common side effects include fatigue, cough, nausea, loss of appetite, skin rash, and itching. More serious side effects that occur less frequently include problems with the lungs, intestines, liver, kidneys, thyroid, adrenal glands, pituitary gland, and other organs.5 Serious side effects that cause the immune system to attack normal organs are more common with ipilimumab.5
CAR T-Cells
CAR T-cell therapy is a new way to get immune cells and T-cells to fight cancer. T-cells are taken from your blood and are genetically engineered to produce receptors on their surface called chimeric antigen receptors (CARs). These receptors allow the T-cells to recognize and attach to a specific protein (called an antigen) that is located on the tumor cells. This will allow the body to kill off those cancer cells.2 Currently, there are only 2 approved CAR T-cell therapies. Tisagenlecleucel (Kymriah) is approved for patients up to the age of 25 years who have B-cell precursor acute lymphoblastic leukemia that is not responding to treatment and has relapsed at least twice. Kymriah is also approved for adults with relapsed or refractory large B-cell lymphoma after 2 or more treatments. Axicabtagene ciloleucel (Yescarta) is approved for treatment of relapsed or refractory B-cell lymphoma after 2 or more treatments have failed.3,6 The long-term side effects of these treatments are not known, but there are some short-term side effects, including one called cytokine release syndrome (CRS). CRS is a potentially serious side effect that can result from an overwhelming immune system response and can make people feel as though they have the “worst flu of their life.”6 CRS can cause confusion, low blood pressure, high fever, chills, nausea, fatigue, rapid heart rate, capillary leakage (when fluid and proteins leak out of tiny blood vessels and flow into surrounding tissue, resulting in very low blood pressure), cardiac arrest, heart rhythm abnormalities, heart failure, hypoxia, decrease in kidney function, poor lung function and oxygenation, organ failure, delirium, involuntary muscle twitching, hallucinations, or even unresponsiveness. Deaths have been associated with CRS, and it must be managed at a knowledgeable center that is equipped to deal with these side effects. A drug called tocilizumab (Actemra) is approved for the treatment of CRS; this drug can help decrease symptoms and decreases the severity of CRS.6
Monoclonal Antibodies
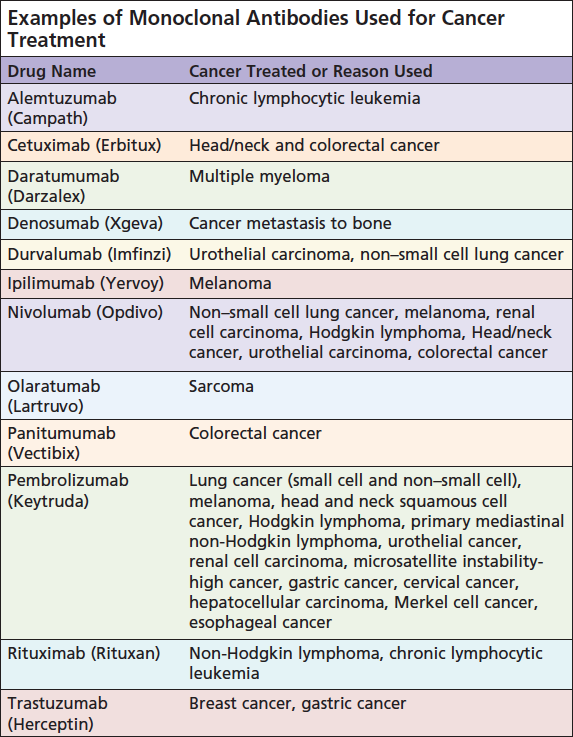
The immune system attacks foreign substances by making large numbers of antibodies. These are proteins that attach to a specific protein on the cell called an antigen.7 Also called therapeutic antibodies, these are produced in the lab. These antibodies are designed to attach to specific antigens found on cancer cells and recruit other cells in the immune system to destroy the cells containing the antigen.7 Some monoclonal antibodies mark the cancer cells so the immune system can more easily recognize them and kill them. Other antibodies are a type of treatment called targeted therapy that does not produce a response from the immune system itself but kills off the specific cells.2 There are different types of monoclonal antibodies, including naked monoclonal antibodies, conjugated monoclonal antibodies, and bispecific monoclonal antibodies.7
Naked monoclonal antibodies are those that work by themselves. There is no drug or other substance attached to the antibody. These are the most common type of antibody used to treat cancer.7 Most naked antibodies attach to antigens on cancer cells themselves, but some of them attach to other noncancerous cells or even proteins not attached to a cell.7 They work in different ways. Some boost the immune system’s own response to cancer cells by attaching to the cancer cell and acting as a marker for the immune system to destroy them. An example of this type of monoclonal antibody is alemtuzumab (Campath), a drug used for the treatment of chronic lymphocytic leukemia.7 Some of the naked antibodies boost the immune system response by targeting checkpoints (described above).7 Some attach to and block antigens on cancer cells and nearby cells that help cancer cells spread and grow. An example of this type of monoclonal antibody is trastuzumab (Herceptin), which is used to treat breast and stomach cancer that has the HER2 protein on its surface.7
Conjugated monoclonal antibodies are antibodies that are joined to a chemotherapy drug or a radioactive particle.7 The monoclonal antibody is used as a homing device to take the chemotherapy or radioactive particle directly to the cancer cell where the antibody delivers the toxic substance into the cancer cell. This lessens the damage to normal cells.7 Conjugated monoclonal antibodies are also called tagged, labeled, or loaded antibodies.
Radiolabeled antibodies have small radioactive particles attached to them; treatment with this type of antibody is often called radioimmunotherapy. An example of a radiolabeled antibody is ibritumomab tiuxetan (Zevalin), which is used for some types of non-Hodgkin lymphoma that have the protein CD20 on their surface.7 Chemolabeled antibodies are monoclonal antibodies that have powerful chemotherapy (or other) drugs attached to them; they are also known as antibody-drug conjugates. The drug used is usually too powerful on its own but can be given safely when attached to an antibody. Examples of chemolabeled antibodies are brentuximab vedotin (Adcetris), which is used to treat Hodgkin lymphoma and anaplastic large cell lymphoma and ado-trastuzumab emtansine (Kadcyla or TDM-1), used to treat breast cancer that has too much of the HER2 protein.7
Bispecific monoclonal antibodies are drugs made up of 2 different monoclonal antibodies; they attach to 2 different proteins at the same time. By attaching to 2 different proteins, this drug can bring cancer cells and the immune system together, which can then cause the immune system to attack the cancer cells. Blinatumomab (Blincyto) is an example of a bispecific antibody used to treat acute lymphocytic leukemia. Blinatumomab attaches to the CD19 protein found on some leukemia and lymphoma cells and to the CD3 protein that is found on T-cells.7
Antibodies are given through a vein (intravenously) or subcutaneously. The antibodies themselves are proteins that can be recognized as foreign substances and can cause an allergic reaction. This is more common when the drug is first given. Other possible side effects include fever, chills, weakness, headache, nausea, vomiting, diarrhea, low blood count, rash, and fatigue.7
Vaccines
Cancer vaccines work by boosting your immune system’s response to cancer cells. These are different from vaccines that are used to prevent a disease. Vaccines are not yet a major type of treatment for cancer but are being studied in clinical trials.8 The immune system is complex, and developing a vaccine to treat cancer is more difficult than many researchers first expected it would be. Researchers are looking at how to best use vaccines and when to give them.8
Other Immune Therapies
Cytokines are chemicals made by some immune system cells. They control growth and activity of immune system cells and blood cells. They can be given by an injection under the skin, into a muscle, or into a vein. Interleukins and IFNs are the most commonly used.
Interleukins are a group of cytokines that act as signals between white blood cells. IL-2 helps the immune system cells grow and divide more quickly. A man-made version of IL-2 is approved for the treatment of kidney cancer. IL-2 can be used as a single therapy or in combination with chemotherapy or other cytokines such as IFNs. IL-2 might be more effective for some cancers when combined with other agents. Side effects include flu-like symptoms such as chills, fever, fatigue, and confusion. Most people gain some weight while receiving IL-2. Nausea, vomiting, or diarrhea may be present. Low blood pressure is common as well but can be treated with other medications. Rare, serious side effects include abnormal heartbeat, chest pain, or other heart problems. IL-2 must be given in a hospital if it is given in high doses.
IFNs are chemicals that help the body resist viral infections and cancer. The types are named after the first 3 letters in the Greek alphabet—IFN-alfa, IFN-beta, IFN-gamma. Only IFN-alfa is used to treat cancer. It can boost the ability of immune cells to attack cancer cells. IFN-alfa may also slow the growth of cancer cells directly, as well as slow the growth of the blood vessels that tumors need to grow. IFN-alfa can be used to treat hairy cell leukemia, chronic myelogenous leukemia, follicular non-Hodgkin lymphoma, cutaneous (skin) T-cell lymphoma, kidney cancer, melanoma, and Kaposi sarcoma. Side effects can include flu-like symptoms such as fever, chills, headache, fatigue, loss of appetite, nausea, and vomiting, and low white blood cell count (which can increase the risk of infection), skin rashes, and thinning hair. Many of the side effects resolve after treatment is completed. However, they can be severe, and treatment may have to be stopped in some patients.
Immunomodulatory drugs such as thalidomide (Thalomid), lenalidomide (Revlimid), and pomalidomide (Pomalyst) are thought to boost the function of the immune system. The exact way they increase the effectiveness is not known. These drugs are mainly used to treat multiple myeloma and other similar cancers that affect plasma cells. Side effects can include drowsiness, fatigue, constipation, diarrhea, low blood cell count, and neuropathy. These drugs can also increase the risk of blood clots and can cause severe birth defects if taken during pregnancy. They also require registration in a Risk Evaluation and Mitigation Strategy program and are very expensive.9
Conclusion/ Take-Home Points
Immunotherapy is becoming much more popular and much more effective treatment for many cancers. Further research into understanding the immune system and development of immunotherapy medications is changing the way cancer is treated. More and more immunotherapy medications are being approved yearly, and the addition of these treatments is expanding. It’s important to understand how these medications work to treat your cancer and what side effects are expected, especially because the side effects can be very different with immunotherapy than they are with chemotherapy.
Reporting side effects quickly will decrease long-term effects. There are times when the medication needs to be held or stopped for safety reasons, but it’s better to report any side effects than risk long-term effects.
References
- American Cancer Society. 2019. What Is Cancer Immunotherapy? www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/what-is-immunotherapy.html. Accessed May 25, 2019.
- National Cancer Institute, 2019. Immunotherapy to Treat Cancer. www.cancer.gov/about-cancer/treatment/types/immunotherapy#3. Accessed May 24, 2019.
- National Comprehensive Cancer Network. NCCN Practice Guideline in Oncology (NCCN Guidelines). Management of Immunotherapy-Related Toxicities. Version 2.2019. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed May 24, 2019.
- Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy; American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768.
- American Cancer Society. Immune checkpoint inhibitors to treat cancer. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/immune-checkpoint-inhibitors.html. Accessed June 11, 2019.
- Leukemia & Lymphoma Society. Chimeric Antigen Receptor (CAR) T-cell Therapy. www.lls.org/treatment/types-of-treatment/immunotherapy/chimeric-antigen-receptor-car-t-cell-therapy. Accessed May 25, 2019.
- American Cancer Society, 2019. Monoclonal Antibodies to Treat Cancer. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/monoclonal-antibodies.html. Accessed May 30, 2019.
- Leukemia & Lymphoma Society. Vaccine Therapy. www.lls.org/treatment/types-of-treatment/vaccine-therapy. Accessed May 5, 2019.
- America Cancer Society, 2019. Non-specific cancer immunotherapies and adjuvants. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/nonspecific-immunotherapies.html. Accessed May 25, 2019.