To download this article as a PDF, see the link below.
TLG1741
Key Facts About Melanoma
Risk Factors for Melanoma
Early Detection of Melanoma
Diagnosing Melanoma
Melanoma Staging
Navigating Your Care Team
Treatment Options and Decisions
Treatments Other Than Surgery
Side Effects
Clinical Trials
Living and Thriving
Recommended Melanoma Websites
Introduction
Melanoma is a type of skin cancer that begins in skin cells called melanocytes. Melanocytes give our skin its color. Although new treatments have brought hope to many patients, some people may not know what these new treatments are, how they work, or what side effects they may experience from these treatments. We believe that having information not only empowers patients and their families as they navigate their treatment, it also helps to improve outcomes. To help you talk to your healthcare team and decide which treatments are best for you, this publication will review melanoma and its treatments. In particular, we will review 2 new classes of drugs that have revolutionized the treatment of melanoma—immunotherapies and targeted therapies.
Key Facts About Melanoma
Melanoma oftentimes, but not always, begins in a mole—that is, a cluster of melanocytes. It can start in the skin or in other parts of the body that contain melanocytes, such as the eyes, the mucous membranes, and the intestines. Although melanoma is more common in people with light skin, it can affect anyone—people with dark skin can also develop melanoma. It is estimated that in 2019, more than 96,480 cases (57,220 in men and 39,260 in women) of invasive melanoma will be diagnosed in the United States, and approximately 7230 people will die of melanoma in the same year. In addition, it is predicted that another 95,830 cases of melanoma in situ (noninvasive type) will be diagnosed in 2019.
Melanoma in situ is the earliest stage of the disease, in which the abnormal cells are present only in the outermost layer of the skin called the epidermis. In other words, in patients with melanoma in situ, the abnormal cells have not spread into the dermis, which is the deeper layer of the skin.
Risk Factors for Melanoma
Our genes are our genetic material that make us who we are. When there is a change in a gene, it is called a mutation. Mutations (or changes) in certain genes can increase a person’s risk for melanoma. If someone in your family has had melanoma, pancreatic cancer, astrocytoma (brain cancer), or mesothelioma, that can also increase your risk for developing melanoma. Other risk factors for melanoma include having a lot of moles or moles that do not look normal, skin that burns easily, red hair, and light-colored eyes. A weak immune system (the immune system is in charge of keeping us healthy) and having had cancer as a child can also increase your risk for melanoma.
Although there are some risk factors beyond our control such as the genes we inherit, other types of risk factors can be minimized or controlled. One of the most common risk factors for melanoma is skin damage from ultraviolet (UV) rays. Sunshine has UVA and UVB light, which can damage the skin. Tanning beds and lamps are not any safer than actual sunshine, because they also produce UVA and UVB light. You can protect yourself from UV light by staying out of the sun (and away from tanning beds and lamps), by wearing protective clothing (a hat, long sleeves), and by using sunscreen when outdoors (see Figure 1).1
Early Detection of Melanoma
When detected early, melanoma is highly treatable. This is why it is so important to find melanoma early, even if you have already had melanoma or other skin cancers. Check your skin regularly. Use a mirror or ask for help to examine difficult-to-see areas, such as your back or scalp. Even though darker-skinned people are less likely to experience sunburn, they can still develop melanoma, which most often begins on the palms of the hands, the soles of the feet, or under the nails in these individuals. Make sure to check these areas, too.
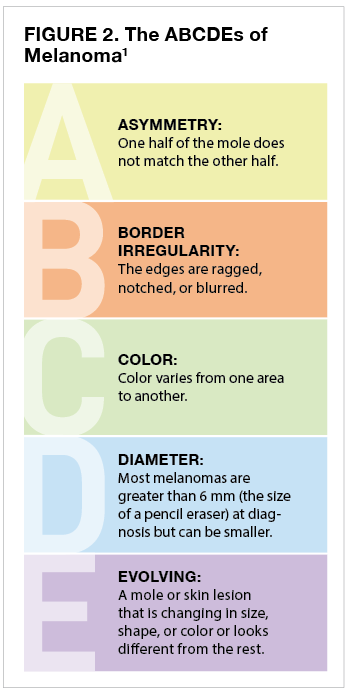
Keep track of the spots on your skin and make note of any changes. It also helps to take pictures if you should ever need to show your doctor how the spot or lesion on your skin is changing. If you notice a mole that looks different from the others, that changes, that itches, or that bleeds, you should make an appointment with a dermatologist (skin specialist) as soon as possible. Patients who have had melanoma should see their dermatologist at least once every year. See Figure 2 for guidance on what to look for in detecting possible melanoma.1
Diagnosing Melanoma
Often, but not always, patients say that their melanoma started out as a dark-colored mole. As it grows into the deeper layers of the skin, however, melanoma can spread to the lymph nodes and even to other organs of the body.
To determine whether you have melanoma, a doctor will begin by removing all or part of the suspicious spot on your skin. This is called a biopsy. Usually, the biopsy is an “excisional” biopsy, in which all of the suspicious skin lesion is removed to make a proper diagnosis.2 This is different from an “incisional” biopsy, in which only part of the lesion is removed for diagnosis.3 The excisional biopsy is usually done either in the shape of an ellipse (like a flat football, to make it easier to close the skin), by shaving the melanoma from the skin’s surface or through a punch biopsy (using a sharp circular tool to take a deeper sample).4 Before you undergo the biopsy, your healthcare provider will numb the area. A pathologist (a doctor who has special training in identifying diseases by studying cells and tissues under a microscope) will then examine the biopsy (the skin and the underlying tissue removed by your doctor) and prepare a pathology report in approximately 3 to 7 days. The report will tell you the following:
- Whether melanoma is present
- The thickness of the melanoma (ie, how deep does the melanoma grow into the skin layers)
- Whether ulceration (tissue breakdown) is present
- Whether immune cells (a certain type of white blood cells called lymphocytes) are present in the melanoma
- Whether any melanoma is present at the edge and the deepest portion of the biopsy (ie, margin status).
Almost 70% of all melanomas are the superficial spreading type; these usually begin in a mole that is already present. Approximately 15% of all melanomas are nodular melanomas; these usually appear rapidly as a black skin bump. Lentigo maligna melanoma usually occurs in older people and on skin that receives a lot of sun exposure, such as the face, ears, and arms. More rare types of melanoma are acral, ocular, and mucosal. Acral melanoma is a kind of melanoma that occurs on the palms of the hands and the soles of the feet. Ocular melanoma is melanoma of the eye. Mucosal melanoma is a melanoma that occurs in the mucosal surfaces of the body such as the mouth, nasal passages, vagina, anus, and other areas. This type of melanoma is not related to or affected by UV light exposure.
Melanoma Staging
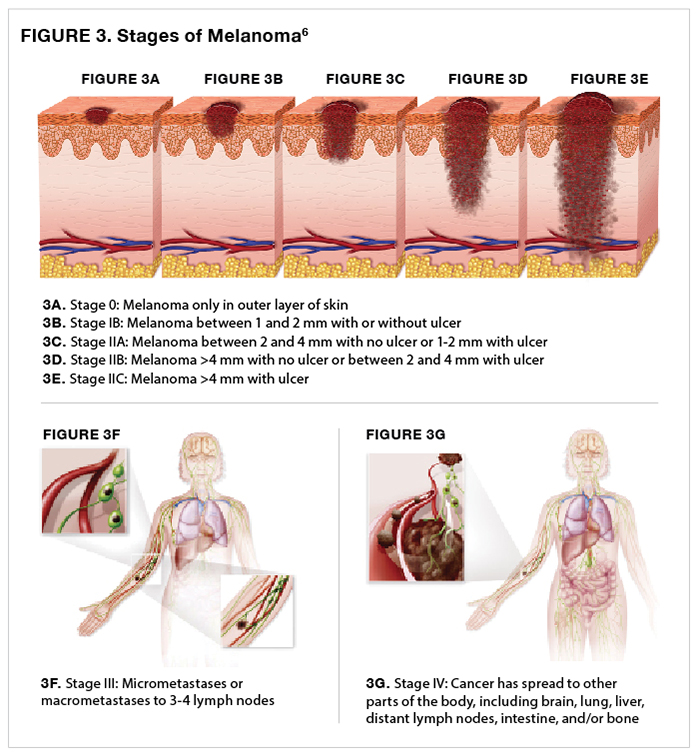
Staging defines the “extent of cancer in the body,”5 taking into consideration the depth of the melanoma, whether it has ulcerated, whether it has spread, and if so, to where and to what extent (Figure 3).6
Navigating Your Care Team
Most melanomas are diagnosed by a dermatologist, who will closely examine your entire body and biopsy any suspicious-looking skin lesions. A pathologist will examine the biopsy to see if you have melanoma and, if so, to stage the melanoma. If you have a high-risk melanoma, you may be sent to a doctor who specializes in cancer surgery and treatment, known as a surgical oncologist. Your doctor will talk to you about the next steps, including, how wide the excision needs to be and whether it will be necessary to remove nearby lymph nodes. If the defect from the melanoma removal is expected to be large or on the face, you may be referred to a plastic surgeon who will assist with closing the wound. A plastic surgeon is an expert in closing the defect created from surgery, to achieve the best possible cosmetic results.
Because melanoma treatment can be complex and dependent on your stage, several other types of doctors may also be involved in your care. If you have a stage III or IV melanoma, a radiation oncologist and a medical oncologist should be part of your treatment team, so that they can talk with you about your treatment choices both before and after surgery. A radiation oncologist uses radiation to treat patients. The use of radiation at the site where the melanoma is removed can help to rid the body of any remaining cancer cells and reduce the risk for cancer recurrence. Medical oncologists can use medications, such as immunotherapies and targeted therapies, to treat the melanoma.
Other clinical team members may include counselors, social workers, nurses, therapists, dietitians, and pharmacists. If you do not have health insurance or need help understanding your out-of-pocket medical expenses, ask to meet with a financial counselor and/or social worker. You can also request to see a nurse or patient navigator; this individual will listen, answer questions, and help you communicate with your healthcare team, so you can schedule your appointments and receive the care that you need.
Figure 4 shows various individuals who may be part of your clinical care team.
It is also important to reach out to family, friends, and your community for support. Your support network can help you get to and from appointments, keep up with such everyday tasks as cooking and cleaning, accompany you to your appointments to listen to and remember what was said, and take notes that you can read later. You should start a notebook and a folder to help keep you organized and request a copy of your cancer pathology report for your personal files. Should you end up having cancer surgery or radiation, keeping a copy of your surgical procedure note and a summary of your radiation treatment in your folder is also recommended.
The following is a list of questions you may want to have available at your next doctor’s appointment:
- What type or subtype and stage of melanoma do I have?
- Can you explain my pathology report to me?
- Has my melanoma spread?
- What are my treatment choices and which plan do you recommend?
- Do I need a sentinel node biopsy? (It is believed that if the cancer has spread, the first place it would likely spread to is the nearby draining lymph node, which is called the sentinel lymph node.)
- What is the goal of the recommended treatment? Is it to get rid of the cancer, help me feel better, or both?
- What are the side effects of the recommended treatment? Can anything be done so that I do not experience these side effects, or what can we do to deal with them?
- How will each treatment affect my everyday life? Will I be able to work, exercise, and perform my usual activities?
- Are there clinical trials available for my type of melanoma?
- What support services are there for my family and for myself?
- Who can help me pay for my cancer treatment?
Treatment Options and Decisions
Surgery
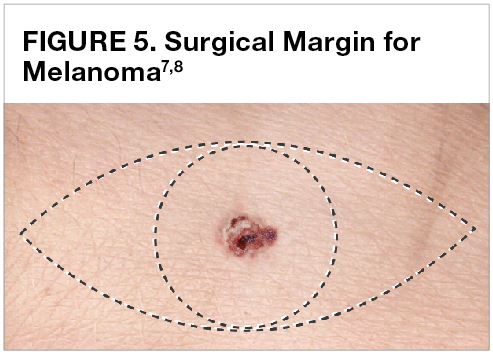
The primary treatment for melanoma is surgical removal, with the thickness of melanoma dictating the extent of surgery. Not only will your surgeon remove the tumor, but he or she will also remove a margin of normal skin along with the tumor, this is known as the surgical margin (Figure 5).7,8
This margin is necessary because some tumor cells may be present in what appears to be normal tissue. Removing an adequate margin helps decrease the chances of the tumor coming back. The amount of skin and underlying tissue that is removed depends, in large part, on how deep the melanoma has penetrated your skin. Thicker tumors require wider surgical margins. For example, the recommended surgical margin for a melanoma that is less than 1 mm deep is 1 cm; whereas the surgical margin for a tumor that is more than 1 mm deep is 2.0 cm. Because melanoma in situ is limited to the topmost layer of the skin, it requires an even smaller margin of 0.1 to 0.5 cm.9 After surgery, the pathologist will look for cancer at the surgical margins. If cancer is found at the margins, you may need to undergo additional surgery to remove any remaining cancer.
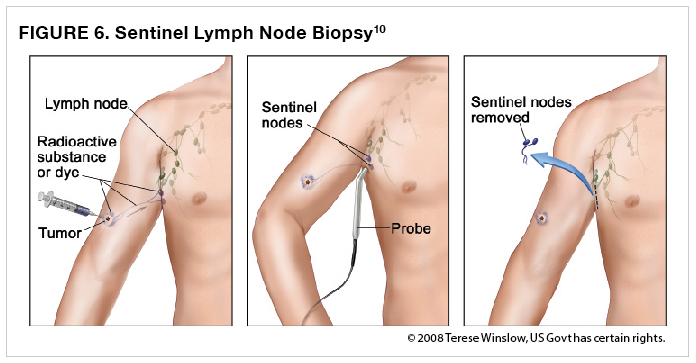
In addition to your primary melanoma being removed, you may also need to undergo surgery on your lymph node(s). The thought is that if the cancer has spread, the first place it would likely spread is to the sentinel lymph node. The deeper the melanoma, the more likely it is to spread to the lymph node. Therefore, the depth of melanoma is utilized in arriving at the decision to perform a sentinel lymph node biopsy. A sentinel lymph node biopsy is performed when there is a high enough likelihood that the melanoma has spread to the lymph nodes, but no abnormal lymph nodes were found during the physical examination. A sentinel lymph node biopsy is usually not performed when it is highly unlikely that the cancer has spread to the lymph nodes—this is generally the case with melanomas that are less than 1 mm deep. Your doctor will find out which node is the sentinel node by injecting a dye close to the tumor. He or she will then follow the path of the dye throughout your lymphatic system to the first node it reaches—that is, the sentinel lymph node—and remove that node (Figure 6).10
A pathologist can then examine the sentinel lymph node with a microscope for the presence of melanoma cancer cells. The sentinel lymph node is usually removed at the same time as the primary melanoma, so additional surgery is not necessary.9
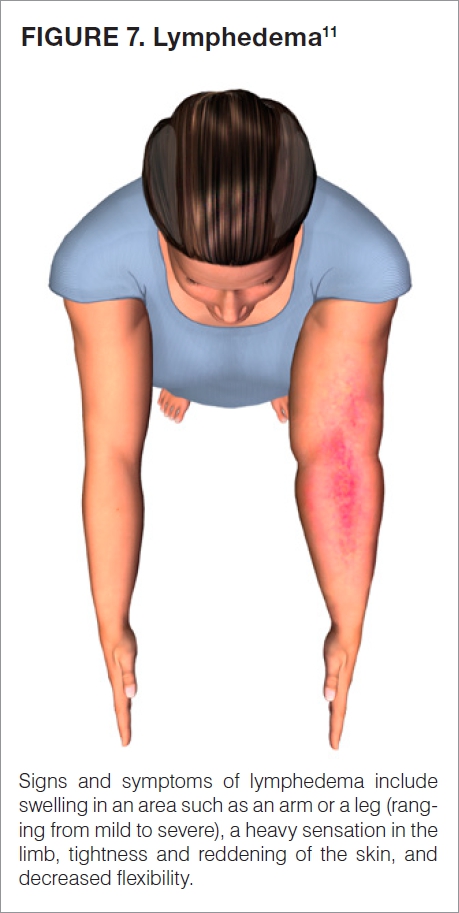
Following removal of your sentinel lymph node, you may experience pain, numbness, bruising, and/or a buildup of lymphatic fluid, which is known as a lymphocele. Lymphedema is a possible, more serious side effect, in which the arm or leg swells because of a backup of lymphatic fluid (Figure 7).11
Lymphatic fluid can build up when the lymphatic vessels are clogged, damaged, or have been surgically removed.12 Lymphedema does not usually happen when only a few lymph nodes have been removed.9
You may also require a lymph node dissection if your doctor thinks that the cancer has spread to a lymphatic basin—that is, a group of lymph nodes to which a region of the body drains13—perhaps because cancer cells were detected in the sentinel lymph node. In a lymph node dissection, all the lymph nodes in the nodal basin are removed. Lymphedema is common following a lymph node dissection, and it can be permanent. Other common side effects following a lymph node dissection include numbness, pain, and decreased range of motion in your arm or leg.9
Another way to look for cancer cells is to perform a fine-needle aspiration (FNA) biopsy. Unlike a sentinel lymph node biopsy, in which an abnormal lymph node cannot be found during the physical examination, an FNA is performed when an abnormal lymph node can be felt. In an FNA biopsy, the abnormal lymph node is not removed. Instead, a needle is placed into it to remove cells. A pathologist then will examine the tissue with a microscope to check for the presence of any cancerous cells.9
If your doctor feels an abnormal lymph node, but an FNA cannot be performed or did not provide enough information to make a diagnosis, then he or she may need to cut out the abnormal node(s) and some of the surrounding normal tissue. This is known as an excisional lymph node biopsy. You will require local or general anesthesia when this type of surgery is performed.9
Gene Mutations
The genetic changes in cancer cells are called mutations. Certain cancers exhibit specific gene mutations that the cancer depends on for its growth; blocking the action of these genes can help control cancer growth. For example, BRAF gene mutations are found in approximately 50% of melanomas in the skin. NRAS gene mutations are found in approximately 20% of melanomas, and NF-1 gene mutations are present in approximately 10% to 15% of all melanomas. KIT gene mutations are more common in melanomas that begin in mucous membranes, on the hands or feet, or in skin that has been repeatedly damaged by the sun, such as lentigo maligna melanoma. Your melanoma may not have any of these mutations but could still have other genetic changes that cause it to grow. Several medications have recently been developed to interrupt the action of these genes to control cancer growth. Knowing which genes are expressed by your melanoma can help guide your treatment in certain situations, as discussed later in this publication.
Biomarker/Molecular Testing
BRAF mutation testing is recommended for patients with stage III melanoma who are at high risk for their melanoma recurring or for whom knowing their BRAF status may change their course of treatment.14 When patients have clinical recurrence or stage IV disease at diagnosis, they should be tested for BRAF and KIT mutations if targeted therapy is an option. Additional testing, including BRAF non-V600 mutations and an expanded next-generation sequencing (NGS) panel, may also be performed if the results will help select a treatment or determine whether a patient qualifies for a clinical trial. (NGS refers to the sequencing of nucleotides, which are the building blocks of DNA and RNA.15) Additional NGS testing to look for other gene mutations that may be possible targets for treatment may also be appropriate if the initial BRAF single-gene testing is negative.14
Treatments Other Than Surgery
Sometimes patients with melanoma will receive treatments in addition to, or instead of, surgery. When another treatment is administered after the primary treatment (which is usually surgery), this is called adjuvant treatment. Patients with melanoma stages 0, IA, IB, and IIA do not require adjuvant therapy because of the low risk for recurrence; patients with stage III melanoma may be eligible for adjuvant therapy and should discuss this option with their healthcare team.9 Treatments other than surgery include immunotherapies, targeted therapies, radiation, and chemotherapies (rarely used). With the treatment paradigm for melanoma rapidly evolving, certain patients with more advanced disease may be appropriate candidates to receive immunotherapy or targeted therapy before surgery. This type of treatment is called neoadjuvant treatment.
Immunotherapies
Immunotherapies improve the body’s natural ability (ie, the immune system) to fight diseases, including cancer.9 Some immunotherapies work locally, and others work systemically (ie, they affect cells throughout the body). Systemic melanoma immunotherapies include the cytokines interferon (IFN) alfa-2b and aldesleukin (also known as interleukin-2 [IL-2]); the programmed cell death protein 1 (PD-1) inhibitors pembrolizumab and nivolumab; and the cytotoxic T-lymphocyte antigen 4 (CTLA-4)-blocking antibody ipilimumab. Both PD-1 inhibitors and CTLA-4–blocking antibodies belong to the class of drugs known as checkpoint inhibitors. Immunotherapies used as local treatment for melanoma include IL-2, IFN alfa-2b, imiquimod, Bacillus Calmette-Guérin (BCG) Live, and talimogene laherparepvec (T-VEC).9
Cytokines
IL-2 and IFN alfa are both cytokines. Cytokines are normally found in the body, with their job being to activate our immune response. When they are used to treat cancer, they are administered in quantities that are much larger than those found naturally in the body, which can sometimes lead to severe side effects. Both IL-2 and IFN alfa can make you feel as if you have the flu, with muscle aches, headache, fever, chills, and fatigue being commonly reported side effects. IFN alfa can also make you lose your appetite; feel depressed; and can cause hair thinning, vomiting, nausea, and liver damage. IL-2 can also make you feel nauseated and cause you to throw up; can make it difficult to catch your breath; and can cause confusion, heart damage, a skin rash, abnormal liver and/or kidney laboratory values, and fluid buildup.9 IFN alfa and IL-2 are no longer recommended as adjuvant treatments for melanoma of the skin, because the newer targeted therapies and immune checkpoint inhibitors are more effective and are associated with fewer side effects.14
CTLA-4–blocking antibody
Ipilimumab is a CTLA-4–blocking antibody.16 The agent is approved for use in adults and in children 12 years of age or older to treat melanoma that has spread (metastatic) or cannot be removed by surgery (unresectable).17 It can also be used as an adjuvant therapy after the melanoma has been surgically removed, to kill any remaining cancer cells.9 In addition, ipilimumab can be combined with a PD-1 inhibitor to treat patients with advanced melanoma.9
The most common side effects associated with the use of ipilimumab include diarrhea, fatigue, and a rash or itchy skin. Ipilimumab use can also be linked to such serious side effects as intestinal inflammation (colitis); skin blisters and mouth sores; difficulties with the adrenal, thyroid, or pituitary glands; kidney inflammation or failure; vision changes; hepatitis; severe nerve problems, which may lead to paralysis; inflammation of the brain (encephalitis); lung inflammation; severe infusion reactions; and eye problems.9,17
PD-1 inhibitors
Pembrolizumab and nivolumab are PD-1 inhibitors. These drugs activate the body’s natural immunity to foreign invaders, including cancer cells. Pembrolizumab and nivolumab can be used to treat advanced melanoma (melanoma that has metastasized or cannot be surgically removed). These agents can also be used as an adjuvant treatment (to reduce the risk of melanoma recurrence following surgical removal of the melanoma and any cancerous lymph nodes).18,19
Common side effects associated with the use of pembrolizumab and nivolumab include nausea, vomiting, diarrhea, a rash or itchy skin, fatigue, and pain in the joints, muscles, and/or bones.9 Both drugs can also be associated with serious side effects and can “cause your immune system to attack normal organs and tissues in any area of your body.”18,19
Local therapies
Imiquimod, T-VEC, and BCG are 3 local therapies used to treat melanoma. Imiquimod is an immune response modifier that is dispensed as a cream. When surgery is not an option because of a patient’s health or for cosmetic reasons, topical imiquimod can be used, particularly for individuals with lentigo maligna. Imiquimod can also be used before a surgical excision or after an excision that did not remove all of the melanoma or did not leave a large enough tumor-free margin.14
Side effects associated with imiquimod use include skin changes, such as redness, flaking, and stinging, as well as more general symptoms, including fatigue, diarrhea, headache, or back pain. The use of imiquimod can also be linked to more serious side effects, such as flu-like symptoms and sores.20
T-VEC and BCG are treatments that are injected into the tumor (that is, an intralesional injection). T-VEC is a weakened form of the virus that causes cold sores—herpes simplex virus type 1.21 The injection is usually administered at a hospital or at a clinic. When T-VEC is injected into tumors that have spread (metastasized), it kills melanoma cells at the site where it is injected, triggers a nearby immune response, and sometimes kills melanoma cells that are elsewhere in the body.9
The most common side effects associated with T-VEC use are nausea, vomiting, fever, chills, fatigue, a flu-like illness, and injection-site pain; other side effects include cellulitis and swelling.14 BCG is made from Mycobacterium bovis—a bacterium that has been weakened.22 Once it is injected into the tumor, BCG stimulates the immune system to attack the melanoma cells.9 BCG is not used clinically very much anymore—it is associated with severe local adverse effects and occasionally with bodywide adverse effects. In a study by Krown and colleagues, local reactions included skin redness and ulceration. Systemic reactions included headache, fever, and chills, as well as less frequent episodes of low blood pressure, nausea, and vomiting.23
Vaccines
Scientists are currently researching the safety and efficacy of vaccines in clinical trials for the treatment of melanoma.9
Targeted Therapies
In addition to immunotherapies, another class of drugs—targeted therapies—is available to treat melanoma. These agents are designed to attack cancer cells while minimizing the damage to normal cells, so that patients experience fewer side effects. To do this, some targeted therapies interfere with the molecules, proteins, or enzymes that the cancer needs to grow or spread. Some targeted therapies kill the cancer cells by bringing toxic material directly to them, whereas others combat cancer by helping the immune system to zero in on it.24
Vemurafenib, dabrafenib, and encorafenib are BRAF inhibitors—a type of targeted therapy known as signal transduction inhibitors.14 The MEK inhibitors—cobimetinib, trametinib, and binimetinib—are also signal transduction inhibitors.14,25 Combination therapy with a BRAF inhibitor plus an MEK inhibitor can have a significant impact on the treatment of melanoma compared with the use of either agent alone.25 These drugs target cancer cells with specific mutations to the BRAF gene; they do not work in melanomas that do not have the BRAF mutation.9
Imatinib mesylate is a signal transduction inhibitor that targets cells with a mutation to the c-kit gene. In patients with melanoma, a mutated c-kit gene occurs less often than a mutated BRAF gene.9
The most common side effects associated with use of the BRAF inhibitors vemurafenib, dabrafenib, and encorafenib include rash, sun sensitivity, thickened skin, nausea, fever, headache, fatigue, hair loss, and joint pain. More serious, but less common side effects are a severe allergic reaction, kidney failure, skin and liver problems, bleeding, and increased blood sugar levels. Finally, squamous-cell carcinoma could develop.9,26,27 You should tell your doctor about any changes to your skin, and he or she should regularly check your skin while you are receiving treatment and for several months thereafter.27
The most common side effects associated with use of the MEK inhibitors cobimetinib, trametinib, and binimetinib include nausea, diarrhea, swelling, rash, and sun sensitivity. Lung, liver, heart, and muscle damage, eye problems, bleeding or blood clots, and skin infections are severe, less common side effects that could develop.27
Side effects associated with imatinib include swelling, nausea, vomiting, diarrhea, loss of appetite, fatigue, muscle or bone pain, and rash. Imatinib is also associated with more serious side effects, such as heart and liver problems, increased pressure in the brain or heart, abnormal bleeding, skin blistering, and low levels of certain blood cells and thyroid hormone.28
Chemotherapy
Chemotherapy kills rapidly dividing cells. Cancer cells are rapidly dividing cells, but so are some normal cells. Chemotherapy is used less often nowadays, having been replaced by improved treatments such as immunotherapies and targeted therapies.
Radiation
Radiation can kill cancer cells and stop new cancer cells from growing by damaging their DNA. In patients with melanoma, radiation is infrequently used after surgery (adjuvant treatment) to prevent recurrence of the cancer. The decision regarding whether to use radiation depends on the risk of the melanoma recurring if radiation therapy is not used. Examples of when adjuvant radiation might be used include regional melanoma in which lymph nodes were removed, or certain cases of desmoplastic melanoma (an uncommon malignant melanoma that is not pigmented and is usually found on the head and neck).29
Radiation treatment can also be used palliatively, which means that it is used to control symptoms. Palliative radiation can control the pain and other symptoms patients might experience when melanoma spreads or when surgery is not an option. Radiation therapy may be the primary therapy in certain situations, such as lentigo maligna and brain metastases. Stereotactic radiosurgery can give a high radiation dose to a pinpoint area. For primary treatment, stereotactic radiosurgery/fractionated stereotactic radiation therapy are generally preferred over whole brain radiation therapy.14 Side effects associated with radiation include swelling of the area that was radiated, fatigue, skin changes that resemble a sunburn, and development of a second cancer many years after receiving radiation.9
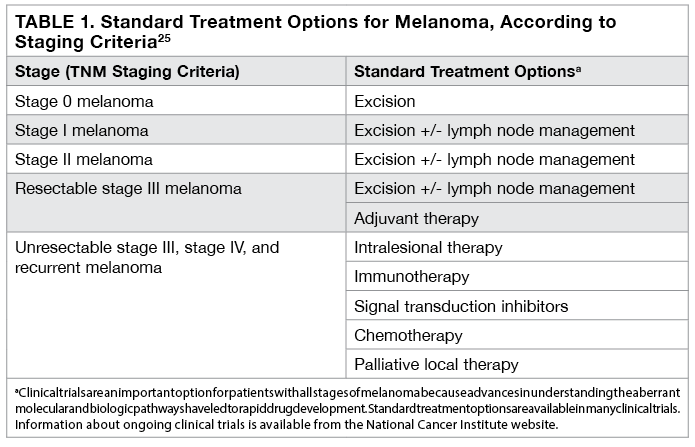
Table 1 provides an overview of the types of treatments for melanoma based on staging criteria.25
Side Effects
Ask your doctor for a list of possible side effects specific to the medication you are taking (see Table 2 for more information on adverse events associated with specific treatments for melanoma).9,14,25-28
Pay attention to any possible side effects and report them during and after completing treatment.
Although immune-related adverse events do not occur very often, they are serious and can quickly become dangerous. Seek medical attention right away if you experience a high fever, difficulty breathing, intense abdominal pain, swelling, or inflammation (this is not every possible symptom).6
The following list of immunotherapy-related side effects and how they might be treated is adapted from the website of the Society for Immunotherapy of Cancer,6 but is not a substitute for medical care.
Constipation – Please discuss any constipation with your doctor so that it does not become a major problem.
Cytokine release syndrome – A rash, rapid heartbeat, headache, fever, nausea, difficulty breathing, and low blood pressure are all symptoms. Cytokine release syndrome is an immune-related adverse event that can be mild to life-threatening.
Diarrhea – If you have diarrhea that keeps you from leaving the house, severe abdominal cramping, or 3 more bowel movements than what is normal for you in a day, call your doctor right away.
Fatigue – Try to prioritize activities and pace yourself.
Flu-like symptoms – Fever, chills, nausea, vomiting, headaches, aches, sleepiness, runny nose, decreased appetite, and blood pressure changes are all possible flu-like symptoms.
Heart palpitations – If you feel dizzy, faint, or as if your heart is not beating normally, call your doctor right away.
Infusion-related reactions – Symptoms include fever, chills, a rash, and itching. With more severe reactions, you might feel as if your heart is not beating normally. You might also experience low blood pressure, dizziness, difficulty breathing, and shaking. Depending on how severe the symptoms, your doctor might want to slow the infusion; give you an antihistamine, steroid, or analgesic; or stop the infusion.
Injection-site reactions – Talk to your doctor about how to manage possible pain or discomfort.
Mouth sores – Report any symptoms to your doctor right away, since mouth sores can be managed when they are caught early.
Muscle and joint pain – Let your doctor know if your pain becomes severe or doesn’t go away once the treatment is finished. The pain might occur in one area or in your whole body.
Nausea and vomiting – There may be medications that can help.
Respiratory symptoms – Tell your doctor right away if you have difficulty breathing or a cough.
Skin reactions – Let your doctor know right away about any concerning changes. Your doctor may recommend an antihistamine, steroid, antibiotic, and/or numbing cream.
Swelling in the legs – Notify your doctor if you have any signs of water retention, such as a heavy or stiff feeling in your legs, swelling, or weight gain.
Clinical Trials
Clinical trials help us to better understand and treat different types of melanoma. Participating in a clinical trial has both benefits and risks. The benefits include the fact that you will have access to new treatments, your participation will help to advance medical knowledge, and your treatment and how you and your cancer respond to the therapy will be followed closely. Of course, clinical trials also have risks. You might experience side effects from the treatment and/or the treatment might not work. In addition, you will need to devote your time to participate in the trial.9
Clinical Trial Phases
There are 4 clinical trial phases that are intended to see how well a treatment works and how safe it is (see Figure 8).
Phase 1 trials focus on safety and dosage, and generally enroll between 20 and 100 healthy volunteers or people with the disease or condition being studied. Phase 1 clinical trials determine how a drug affects the body, including what side effects occur with increasing doses. In short, the purpose of phase 1 clinical trials is to better understand how to minimize risks and maximize benefits. Phase 2 trials focus on side effects and how well a treatment works. In phase 2 trials, up to several hundred participants with the disease or condition can be enrolled. Phase 3 trials compare the treatment being studied with the standard of care and can enroll between 300 and 3,000 participants with the disease or condition. Once a drug has been approved by the US Food and Drug Administration (FDA), it can enter phase 4 trials. This is the point at which the FDA gathers more information regarding safety in clinical use in real-world settings. Several thousand volunteers with the disease or condition being explored can enter phase 4 trials.9,30 If you are interested in participating in a clinical trial, you will need to review, understand, and sign an informed consent, which thoroughly describes the study. In addition, patients must meet certain requirements to participate; this minimizes the impact patient differences might have on the results of the trial.9
Living and Thriving
Living with cancer begins on the day you are told you have melanoma and continues throughout your treatment journey. Treatment may end with surgery, or you may need more treatments to decrease the chances of the melanoma recurring and to make your side effects more bearable. You and your healthcare team are on a level playing field, with decision-making being shared. Living with cancer is personal and may include a wide range of feelings, including worry, fear, relief, joy, and hope. Sometimes people feel relieved at the end of treatment. Other times, they may miss their healthcare team’s support and become anxious when they have fewer appointments. You may want to join a support group or participate in community events—sharing your experiences and emotions with people who have gone through similar things can be very comforting and improve your ability to cope. One-on-one counseling with a mental health professional or trained spiritual counselor may also help.
Spouses, family members, and caregivers may also go through a transition period following treatment. The “new normal” is a change for both caregivers and patients, with everyone experiencing this phase differently. Caregivers are often so focused on taking care of the patient that they forget about themselves. It is also important for them to take care of their physical and emotional needs as they cope with having a loved one with cancer—healthy lifestyle choices are good for the patient and for the caregiver as well. Eating healthy, exercising regularly, not smoking, drinking alcohol in moderation, managing stress, and, most importantly, protecting the skin from sun damage are all important tips to follow.
Follow-up care is essential and will include annual skin examinations with a dermatologist, along with other tests based on the type and stage of your cancer. There are assistance programs for people who cannot afford their medications or have unpaid medical bills and living expenses. Reach out to your support team of navigator, financial counselor, and social worker to learn how your institution can help.
Recommended Melanoma Websites
Make sure to use reliable sources, including websites, books, and pamphlets, when you are seeking information about melanoma. A good place to begin is with the following trustworthy websites:
- www.cancer.net - The American Society of Clinical Oncology provides education and resources for people living with cancer, including those with melanoma.
- https://medlineplus.gov/melanoma.html - The US National Library of Medicine provides up-to-date, science-based information in English and Spanish, along with links to other sites.
- www.cancer.gov - The National Cancer Institute (NCI) conducts and supports cancer research and training. Use this site to find patient and caregiver resources, as well as links to NCI-supported clinical trials.
- www.patientadvocate.org - The mission of the Patient Advocate Foundation is to provide “case management services and financial aid to Americans with chronic, life threatening and debilitating illnesses.” Start here when you need financial assistance.
- www.cancercare.org - CancerCare provides free emotional and practical support for people with cancer and their caregivers. Oncology social workers offer individual and group support (by phone, online, or in person in New York and New Jersey) in English and Spanish. Financial assistance is also available.
- www.aad.org/public - The American Academy of Dermatology/American Academy of Dermatology Association provides free educational handouts, videos, and other information on skin cancer screening, detection, and intervention.
- www.aimatmelanoma.org – The AIM at Melanoma Foundation is an advocacy organization that offers a peer connect program and toll-free phone number to speak with a healthcare professional about melanoma diagnosis and treatment. An educational video library and patient resource list provide information on reducing your risk of, being diagnosed with, and living with melanoma.
- https://melanomainternational.org - The Melanoma International Foundation is an advocacy organization that provides one-on-one support through e-mail and open discussion forums. The site delivers information “to develop personalized strategies with patients so they may live longer, better lives.”
- https://melanoma.org - The Melanoma Research Foundation supports research, provides information, and advocates for patients with the disease. The organization has resources for pediatric and young adult melanoma survivors, as well as for people with less common types of melanoma (ocular, mucosal).
- www.ocularmelanoma.org - The Ocular Melanoma Foundation focuses on ocular melanoma and provides financial assistance to qualifying patients with ocular melanoma for travel and prosthetics. A patient forum and support events are also available.
References
- American Academy of Dermatology/American Academy of Dermatology Association. Infographic: skin cancer body mole map.www.aad.org/public/spot-skin-cancer/free-resource. Accessed December 12, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Excisional biopsy. www.cancer.gov/publications/dictionaries/cancer-terms/def/excisional-biopsy. Published February 2, 2011. Accessed December 12, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Incisional biopsy. www.cancer.gov/publications/dictionaries/cancer-terms/def/incisional-biopsy. Published February 2, 2011. Accessed December 12, 2019.
- Swetter SM, Tsao H, Bichakjian CK, et al; Work Group. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Staging system. www.cancer.gov/publications/dictionaries/cancer-terms/def/staging-system. Published February 2, 2011. Accessed December 16, 2019.
- Society for Immunotherapy of Cancer (SITC) connectED. Staging Melanoma. www.sitcancer.org/clinician/resources/melanoma/staging. Accessed December 16, 2019.
- Patient Resource Publishing. Melanoma: Surgical treatment. www.patientresource.com/Melanoma_Surgical_Treatment.aspx. Accessed December 17, 2019.
- Nucleus Medical Media. Melanoma excision. https://catalog.nucleusmedicalmedia.com/melanoma-excision/view-item?ItemID=10378. Accessed December 17, 2019.
- National Comprehensive Cancer Network. NCCN Guidelines for Patients®. Melanoma. 2018. www.nccn.org/patients/guidelines/melanoma/index.html. Accessed December 17, 2019.
- National Cancer Institute. Extensive Lymph Node Surgery Does Not Increase Survival in Melanoma. www.cancer.gov/news-events/cancer-currents-blog/2017/lymph-node-surgery-melanoma. Published June 30, 2017. Accessed December 17, 2019.
- Balch CM, Thompson JF, eds. Melanoma: A Treatment Guide for Patients and Their Families. 4th ed. Kansas City, MO: Patient Resource Publishing; 2015. www.patientresource.com/userfiles/file/Melanoma2015.pdf. Accessed December 17, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Lymphedema. www.cancer.gov/publications/dictionaries/cancer-terms/def/lymphedema. Published February 2, 2011. Accessed December 17, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Lymphatic basin. www.cancer.gov/publications/dictionaries/cancer-terms/def/lymphatic-basin. Published February 2, 2011. Accessed December 17, 2019.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Cutaneous Melanoma, Version 3.2019—October 22, 2019. www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf. Accessed December 16, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Genetics Terms. NGS. www.cancer.gov/publications/dictionaries/genetics-dictionary/def/NGS. Published July 20, 2012. Accessed November 18, 2019.
- YERVOY® [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2019. http://packageinserts.bms.com/pi/pi_yervoy.pdf. Accessed December 17, 2019.
- What is YERVOY? YERVOY® (ipilimumab). Yervoy website. www.yervoy.com/metastatic/what-is-yervoy. Accessed December 17, 2019.
- KEYTRUDA® (pembrolizumab) Indications and Important Safety Information. Keytruda website. www.keytruda.com/important-safety-information/. Accessed December 17, 2019.
- OPDIVO® (nivolumab) Medication Guide. Opdivo website. http://packageinserts.bms.com/medguide/medguide_opdivo.pdf. Accessed December 17, 2019.
- Imiquimod Topical. MedlinePlus website. https://medlineplus.gov/druginfo/meds/a698010.html. Accessed December 17, 2019.
- IMLYGIC® (talimogene laherparepvec) | Patient Information. https://www.imlygic.com. Accessed December 17, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. BCG. www.cancer.gov/publications/dictionaries/cancer-terms/def/BCG. Published February 2, 2011. Accessed December 17, 2019.
- Krown SE, Hilal EY, Pinsky CM, et al. Intralesional injection of the methanol extraction residue of Bacillus Calmette-Guerin (MER) into cutaneous metastases of malignant melanoma. Cancer. 1978;42:2648-2660.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Targeted therapy. www.cancer.gov/publications/dictionaries/cancer-terms/def/targeted-therapy. Published February 2, 2011. Accessed December 18, 2019.
- National Cancer Institute. Melanoma Treatment (PDQ®) – Health Professional Version. www.cancer.gov/types/skin/hp/melanoma-treatment-pdq. Published August 23, 2019. Accessed December 18, 2019.
- Tang N, Ratner D. Managing cutaneous side effects from targeted molecular inhibitors for melanoma and nonmelanoma skin cancer. Dermatol Surg. 2016;42(suppl 1):S40-S48. doi:10.1097/DSS.
- American Cancer Society. Targeted Therapy Drugs for Melanoma Skin Cancer. www.cancer.org/cancer/melanoma-skin-cancer/treating/targeted-therapy.html. Accessed December 18, 2019.
- Understanding GLEEVEC® (imatinib mesylate) Side Effects. Gleevec website. www.us.gleevec.com/side-effects/understanding-side-effects/. Accessed December 18, 2019.
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. Desmoplastic melanoma. www.cancer.gov/publications/dictionaries/cancer-terms/def/desmoplastic-melanoma. Published February 2, 2011. Accessed December 18, 2019.
- US Food and Drug Administration. Step 3: Clinical Research. FDA website. www.fda.gov/patients/drug-development-process/step-3-clinical-research. Published April 18, 2019. Accessed December 18, 2019.